
PFIZER REPORTS RECORD FULL-YEAR 2022 RESULTS AND
PROVIDES FULL-YEAR 2023 FINANCIAL GUIDANCE
▪ Full-Year 2022 Revenues of $100.3 Billion, An All-Time High for Pfizer, Reflecting 30% Operational
Growth
– Excluding Contributions from Paxlovid and Comirnaty
(1)
, Revenues Grew 2% Operationally
▪ Strong Fourth-Quarter 2022 Revenues of $24.3 Billion, Reflecting
13% Operational Growth
– Excluding Contributions from Paxlovid and Comirnaty
(1)
, Revenues Grew 5% Operationally
▪ Full-Year 2022 Reported Diluted EPS
(2)
of $5.47, Up 42% Year-Over-Year, and Adjusted Diluted EPS
(3)
of
$6.58, Up 62% Year-Over-Year, Both of Which Represent All-Time Highs for Pfizer
▪ Fourth-Quarter 2022 Reported Diluted EPS
(2)
of $0.87, Up 48% Year-Over-Year, and Adjusted Diluted EPS
(3)
of $1.14, Up 45% Year-Over-Year
– Includes a $0.32 Benefit from Lower Acquired IPR&D Expenses Compared to Fourth-Quarter 2021
▪ Provides Full-Year 2023 Revenue Guidance
(4)
of $67.0 to $71.0 Billion and Adjusted Diluted EPS
(3)
Guidance of $3.25 to $3.45
– Full-Year 2023 Revenues Excluding COVID-19 Products Expected to Grow 7% to 9% Operationally
Compared to Full-Year 2022
– Full-Year 2023 Revenue Guidance for Comirnaty
(1)
of ~$13.5 Billion and Paxlovid of ~$8 Billion
– Revenues from COVID-19 Products Expected to Grow in 2024 After Reaching a Low Point in 2023 Due
to Significant Government Supply on Hand to Start the Year
– Company Plans to Make Significant Incremental Investments in 2023 to Support Launch Products and
R&D Projects that are Expected to Drive its Long-Term Growth Ambitions
▪ Continues to Make Progress on Pfizer’s Unprecedented Number of Anticipated Launches of New Products
and Indications, Including Recent Regulatory Filing Acceptances for Prevnar 20 Pediatric, its RSV Vaccine
for Older Adults, Etrasimod, and its Pentavalent Meningococcal Vaccine
NEW YORK, NY, Tuesday, January 31, 2023 – Pfizer Inc. (NYSE: PFE) reported exceptional financial results
for fourth-quarter and full-year 2022 and provided 2023 financial guidance
(4)
.
The fourth-quarter 2022 earnings presentation and accompanying prepared remarks from management as well as
the quarterly update to Pfizer’s R&D pipeline can be found at www.pfizer.com.
EXECUTIVE COMMENTARY
Dr. Albert Bourla, Chairman and Chief Executive Officer, stated: “2022 was a record-breaking year for Pfizer, not
only in terms of revenue and earnings per share, which were the highest in our long history, but more importantly,
in terms of the percentage of patients who have a positive perception of Pfizer and the work we do. As proud as
we are about what we have accomplished, our focus is always on what is next. As we turn to 2023, we expect to
once again set records, with potentially the largest number of new product and indication launches that we’ve ever
- 1 -

had in such a short period of time. We believe that the combination of these expected near-term launches,
additional pipeline products that could potentially come to market in the medium-term, and anticipated
contributions from business development, has the potential to set the company up for continued robust growth
through the rest of this decade and beyond.”
David Denton, Chief Financial Officer and Executive Vice President, stated: “I am very pleased with our fourth-
quarter performance, which was highlighted by strong operational growth from Paxlovid, Prevnar 20, Comirnaty,
Vyndaqel and Eliquis, as well as the inclusion of Nurtec ODT/Vydura and Oxbryta. For the full-year, we achieved
revenues of over $100 billion, including 10 medicines or vaccines that generated revenues of more than $1 billion
each, and all of this was accomplished despite operating in an environment in which foreign exchange reduced
our revenues by 7%. Looking forward to 2023, we expect strong topline growth of 7% to 9% excluding our
COVID-19 products and anticipated foreign exchange impacts. We are also increasing our investments behind our
launch products and pipeline in order to help realize our growth goals for 2023 and beyond.”
Results for the fourth-quarter and full-year 2022 and 2021
(5)
are summarized below.
OVERALL RESULTS
($ in millions, except
per share amounts)
Fourth-Quarter Full-Year
2022 2021 Change 2022 2021 Change
Revenues $ 24,290 $ 23,838 2% $ 100,330 $ 81,288 23%
Reported Net Income
(2)
4,995 3,393 47% 31,372 21,979 43%
Reported Diluted EPS
(2)
0.87 0.59 48% 5.47 3.85 42%
Adjusted Income
(3)
6,551 4,543 44% 37,717 23,196 63%
Adjusted Diluted EPS
(3)
1.14 0.79 45% 6.58 4.06 62%
REVENUES
($ in millions)
Fourth-Quarter Full-Year
2022 2021
% Change
2022 2021
% Change
Total Oper. Total Oper.
Global Biopharmaceuticals
Business (Biopharma)
(6)
$ 23,922 $ 23,456 2% 13% $ 98,988 $ 79,557 24% 31%
Primary Care
(6)
17,348 16,225 7% 20% 73,023 52,029 40% 49%
Specialty Care
(6)
3,566 3,989 (11%) (3%) 13,833 15,194 (9%) (4%)
Oncology
(6)
3,007 3,242 (7%) (3%) 12,132 12,333 (2%) 2%
Pfizer CentreOne $ 368 $ 382 (4%) 1% $ 1,342 $ 1,731 (22%) (19%)
TOTAL REVENUES $ 24,290 $ 23,838 2% 13%
$ 100,330
$ 81,288 23% 30%
Beginning in the first quarter of 2022, Pfizer implemented changes to its Adjusted
(3)
financial measures with
respect to acquired in-process research and development (IPR&D) costs and amortization of intangibles. More
- 2 -

information about these changes and their impact on the periods presented can be found in the Non-GAAP
Financial Measure: Adjusted Income section of this press release.
Beginning in the third quarter of 2022, Pfizer has made several organizational changes to further transform its
operations to better leverage its expertise in certain areas and in anticipation of potential future new product or
indication launches. These changes include establishing a new commercial structure within Biopharma focused on
three broad customer groups (primary care, specialty care and oncology)
(6)
, optimizing our end-to-end R&D
operations and further prioritizing our internal R&D portfolio, as well as realigning certain enabling and platform
functions across the organization to ensure alignment with this new operating structure.
Prior period amounts have been revised to conform to the current period presentation for all changes discussed
above.
Business development activities
(7)
completed in 2021 and 2022
(5)
impacted financial results in the periods
presented. Some amounts in this press release may not add due to rounding. All percentages have been calculated
using unrounded amounts. References to operational variances pertain to period-over-period changes that exclude
the impact of foreign exchange rates
(8)
.
2023 FINANCIAL GUIDANCE
(4)
Pfizer’s 2023 financial guidance is presented below. This guidance includes management’s expectations for
contributions from the entire company, including Comirnaty
(1)
and Paxlovid.
2022 Actual Results 2023 Financial Guidance
Revenues $100.3 billion $67.0 to $71.0 billion
Operational
(8)
Growth/(Decline) vs. Prior Year 30% (33%) to (29%)
Growth/(Decline) vs. Prior Year 23% (33%) to (29%)
Adjusted
(3)
Diluted EPS $6.58 $3.25 to $3.45
Operational
(8)
Growth/(Decline) vs. Prior Year 71% (50%) to (47%)
Growth/(Decline) vs. Prior Year 62% (51%) to (48%)
The midpoint of the guidance range for revenues reflects a 31% operational decrease compared to 2022 revenues.
Company revenues are anticipated to be lower in 2023 than in 2022 due entirely to expected revenue declines for
Pfizer’s COVID-19 products.
Excluding COVID-19 products, the Company continues to expect 7% to 9% operational revenue growth in 2023.
Revenue guidance for Pfizer’s COVID-19 products is as follows:
▪ Comirnaty
(1)
revenues of approximately $13.5 billion, down 64% from actual 2022 results.
- 3 -
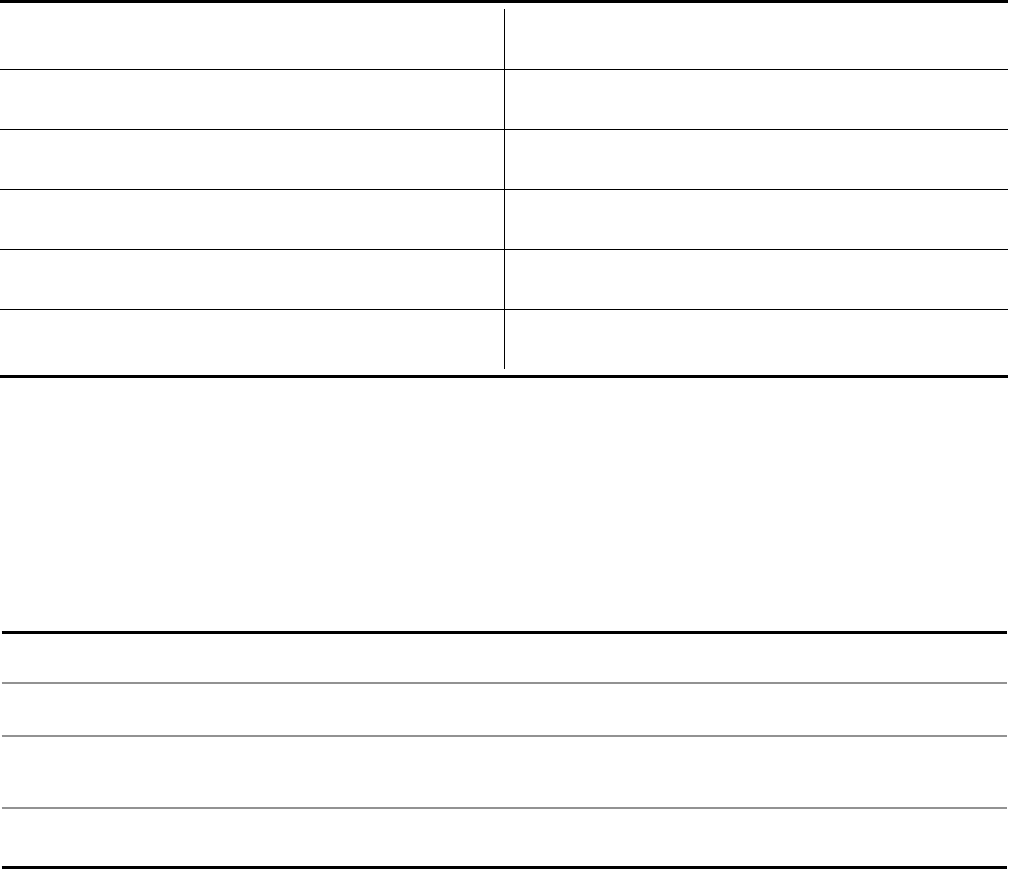
▪ Paxlovid revenues of approximately $8 billion, down 58% from actual 2022 results.
▪ In contrast to previous years, guidance for both products is no longer based primarily on expected deliveries
under existing signed or committed supply contracts, but now also includes, among other things, anticipated
sales through traditional commercial markets in the U.S. in the second half of 2023.
The midpoint of the guidance range for Adjusted
(3)
diluted EPS reflects a 49% operational decrease compared to
2022, primarily driven by anticipated lower revenues from COVID-19 products, higher spending to support
anticipated near-term launches and greater investments in certain late-stage pipeline projects.
Financial guidance for Adjusted diluted EPS
(3)
is calculated using approximately 5.75 billion weighted average
shares outstanding, and assumes no share repurchases in 2023.
Other components of Pfizer’s 2023 financial guidance are presented below.
Adjusted
(3)
Cost of Sales as a Percentage of Revenues 28.0% to 30.0%
Adjusted
(3)
SI&A Expenses $13.8 to $14.8 billion
Adjusted
(3)
R&D Expenses $12.4 to $13.4 billion
Acquired IPR&D Expenses
(4)
Approximately $0.1 billion
Adjusted
(3)
Other (Income)/Deductions Approximately $1.5 billion of income
Effective Tax Rate on Adjusted
(3)
Income Approximately 15.0%
Pfizer’s 2023 financial guidance is based on estimates and assumptions which are subject to significant
uncertainties, particularly with regard to the anticipated performance of Comirnaty
(1)
and Paxlovid, for which
patient demand could be significantly impacted by the infectiousness and severity of the predominant strains of
the SARS-CoV-2 virus during 2023.
Key assumptions incorporated within the guidance follow.
Key Assumptions for 2023 Guidance Commentary
Operational revenue growth compared to 2022
excluding COVID-19 products
7% to 9%
Growth expected to be split among each of three
categories: launch, acquired and in-line products
Incremental SI&A spend to support anticipated
new launches, acquired assets and commercial
launch of COVID-19 products
~$1.3 billion
Investments to support short- and long-term growth
aspirations
Incremental R&D spend to support high-value
pipeline programs and acquired assets
~$1.5 billion
Includes, among others: GLP-1, elranatamab,
respiratory combination vaccines
- 4 -

Comirnaty - 2023 Guidance Assumptions Commentary
Estimated proportion of U.S. population that
receives a vaccine
~24%
Compared to ~31%
†
in 2022; Decrease due to
fewer primary vaccinations and lower compliance
Estimated number of doses per vaccinated person
per year, on average
~1.3 doses
Compared to ~1.4 doses
†
in 2022; Decrease due to
fewer primary vaccinations
Estimated Comirnaty market share - U.S. ~64%
Consistent with share achieved with most recent
bivalent booster in 2022
†
Estimated total demand for Comirnaty doses -
U.S. (includes use of existing government supply)
~65 million
doses
Compared to ~92 million doses
†
in 2022
Assumed timing for delivery of the contracted
doses of Comirnaty to the European Commission
Re-phased over
multiple years
(not all in 2023)
Negotiations on re-phasing of delivery timelines
are ongoing
Paxlovid - 2023 Guidance Assumptions Commentary
Estimated number of total reported symptomatic
infections - global*, excluding China
~112 million
Compared to ~110 million
†
in 2022; Increase due
to expected waning of population immune
protection due to reduced vaccination rates
Estimated proportion of symptomatic COVID-19
patients treated with an oral antiviral treatment -
global*, excluding China
~17%
Compared to ~12%
†
in 2022 (partial year only);
Increase due to greater awareness/education and
full-year implementation
Estimated Paxlovid share of oral antiviral market -
global*, excluding China
~90% Consistent with share achieved in 2022
†
Estimated total demand for Paxlovid - global*,
excluding China (includes use of existing
government supply)
~17 million
courses
Compared to ~12 million courses
†
in 2022 (partial
year only); Increase due to broad product
availability, greater awareness/education and full-
year implementation
Paxlovid sales to China
Assumes no sales
after April 1, 2023
Temporary National Reimbursement Drug List
currently set to end on April 1, 2023
General - 2023 Guidance Assumption Commentary
Estimated timing for transitioning Comirnaty and
Paxlovid to commercial market in the U.S.
Second half of
2023
Assumes prior absorption of existing government
supply
* Only includes markets where Paxlovid is available, and only includes individuals age 12+/18+ where authorized/approved in accordance with local
labeling.
† Actual 2022 market data is derived from a combination of public data sources and internal market research.
CAPITAL ALLOCATION
During full-year 2022, Pfizer deployed its capital in a variety of ways, which primarily include the following two
broad categories:
▪ Reinvesting capital into initiatives intended to enhance the future growth prospects of the company,
including:
▪ $11.4 billion invested in internal research and development projects, and
- 5 -
▪ Approximately $26 billion invested in completed business development transactions, net of cash
acquired, including approximately $12.7 billion
(7)
for the acquisition of Biohaven Pharmaceutical
Holding Company Ltd. (Biohaven), $6.4 billion
(7)
for the acquisition of Arena Pharmaceuticals, Inc. and
approximately $5.6 billion
(7)
for the acquisition of Global Blood Therapeutics, Inc. (GBT).
▪ Returning capital directly to shareholders through a combination of:
▪ $9.0 billion of cash dividends, or $1.60 per share of common stock, and
▪ $2.0 billion, which was used to repurchase 39.1 million shares on the open market in March 2022, at an
average cost of $51.10 per share.
As of January 31, 2023, Pfizer’s remaining share repurchase authorization is $3.3 billion. Current financial
guidance does not anticipate any share repurchases in 2023.
Fourth-quarter 2022 diluted weighted-average shares outstanding used to calculate Reported
(2)
and Adjusted
(3)
diluted EPS were 5,743 million shares, a decrease of 26 million shares compared to the prior-year quarter,
primarily due to shares repurchased in first-quarter 2022, partially offset by shares issued for employee
compensation programs.
QUARTERLY FINANCIAL HIGHLIGHTS (Fourth-Quarter 2022 vs. Fourth-Quarter 2021)
Fourth-quarter 2022 revenues totaled $24.3 billion, an increase of $452 million, or 2%, compared to the prior-year
quarter, reflecting operational growth of $3.0 billion, or 13%, as well as an unfavorable impact of foreign
exchange of $2.5 billion, or 11%. Excluding contributions from Paxlovid and Comirnaty
(1)
, company revenues
grew $571 million, or 5%, operationally.
Fourth-quarter 2022 operational growth was primarily driven by:
▪ Comirnaty
(1)
in developed markets, up 67% operationally, driven primarily by the resumption of deliveries of
the Omicron-adapted bivalent booster following a previously announced period of significantly lower
deliveries of the original vaccine during third-quarter 2022, primarily involving the European Union (EU)
and Japan;
▪ Paxlovid outside the U.S., which contributed $1.8 billion in revenues, driven by international launches in late
2021 and early 2022 following regulatory approvals or emergency use authorizations (EUAs);
▪ Prevnar family (Prevnar 13 & 20) in the U.S., up 79%, driven primarily by strong patient demand following
the launch of Prevnar 20 for the eligible adult population and favorable timing of Centers for Disease Control
and Prevention (CDC) purchasing of the pediatric indication, partially offset by a reduction in revenues due
to a one-time CDC inventory return program for the pediatric indication, the revenue impact of which is
expected to be reversed in 2023 upon replenishment;
- 6 -
▪ Revenues from recently acquired products, Nurtec ODT/Vydura and Oxbryta, which contributed $211
million and $73 million in global revenues, respectively;
▪ Vyndaqel family (Vyndaqel, Vyndamax, Vynmac) globally, up 31% operationally, driven by continued
strong uptake of the transthyretin amyloid cardiomyopathy indication, primarily in developed Europe and the
U.S., partially offset by a planned price decrease that went into effect in Japan in second-quarter 2022;
▪ Eliquis in the U.S., up 17%, driven primarily by continued oral anti-coagulant adoption and market share
gains in non-valvular atrial fibrillation, as well as favorable changes in channel mix; and
▪ Prevenar 13 in emerging markets, up 22% operationally, driven primarily by strong growth in China and
favorable timing of sales to GAVI, the Vaccine Alliance,
partially offset primarily by lower revenues for:
▪ Comirnaty
(1)
in emerging markets, down 81% operationally, primarily due to lower demand for COVID-19
vaccines;
▪ Xeljanz globally, down 28% operationally, driven primarily by declines in net price due to unfavorable
changes in channel mix in the U.S. and decreased prescription volumes globally resulting from ongoing
shifts in prescribing patterns related to label changes;
▪ Sutent globally, down 50% operationally, primarily driven by lower volume demand in Europe following its
loss of exclusivity in January 2022;
▪ Ibrance globally, down 4% operationally, driven primarily by increases in the proportion of patients
accessing Ibrance through the U.S. Patient Assistance Program, planned price decreases that recently went
into effect in international developed markets and prior-year clinical trial purchases internationally, partially
offset by higher volumes across multiple regions; and
▪ Eliquis internationally, down 7% operationally, primarily driven by declines in certain emerging markets.
- 7 -

GAAP Reported
(2)
Income Statement Highlights
SELECTED REPORTED COSTS AND EXPENSES
(2)
($ in millions) Fourth-Quarter Full-Year
2022 2021
% Change
2022 2021
% Change
Total Oper. Total Oper.
Cost of Sales
(2)
$ 9,648 $ 9,736 (1%) 11% $ 34,344 $ 30,821 11% 21%
Percent of Revenues 39.7% 40.8% N/A N/A 34.2% 37.9% N/A N/A
SI&A Expenses
(2)
4,644 4,104 13% 17% 13,677 12,703 8% 11%
R&D Expenses
(2)
3,615 3,445 5% 7% 11,428 10,360 10% 12%
Acquired IPR&D Expenses
(2)
73 2,469 (97%) (97%) 953 3,469 (73%) (73%)
Other (Income)/
Deductions––net
(2)
( 846) ( 835) 1% 13% 217 ( 4,878) * *
Effective Tax Rate on
Reported Income
(2)
4.4% 6.5% 9.6% 7.6%
* Indicates calculation not meaningful.
Fourth-quarter 2022 Cost of Sales
(2)
as a percentage of revenues decreased 1.1 percentage points compared with
the prior-year quarter. The decrease was primarily driven by favorable changes in sales mix, including increased
sales of Paxlovid and higher alliance revenues, as well as favorable impacts resulting from changes in foreign
exchange rates, partially offset by approximately $600 million and approximately $200 million of inventory
write-offs related to Paxlovid and Comirnaty
(1)
, respectively, and higher operational revenues for Comirnaty
(1)
.
SI&A Expenses
(2)
increased 17% operationally compared with the prior-year quarter, primarily reflecting
increased investments to support Paxlovid, Comirnaty
(1)
and recently acquired and launched products.
Fourth-quarter 2022 R&D Expenses
(2)
increased 7% operationally compared with the prior-year quarter, primarily
driven by increased costs to support various vaccine and oncology programs, as well as spending related to
recently acquired assets, partially offset by lower spending on programs to treat COVID-19 and certain other late-
stage clinical programs.
Acquired IPR&D Expenses
(2)
decreased 97% operationally compared with the prior-year quarter. The acquisitions
of Biohaven and GBT in fourth-quarter 2022 qualified as business combinations under U.S. Generally Accepted
Accounting Principles (GAAP), resulting in no Acquired IPR&D Expenses
(2)
, while the acquisition of Trillium
Therapeutics Inc. in fourth-quarter 2021 was accounted for as an asset acquisition, giving rise to approximately
$2.1 billion in Acquired IPR&D Expenses.
Other income––net
(2)
increased 13% operationally in fourth-quarter 2022 compared with fourth-quarter 2021,
primarily driven by net gains on equity securities in fourth-quarter 2022 versus net losses on equity securities
recognized in the prior-year quarter and lower net interest expense, partially offset by lower net periodic benefit
credits associated with pension and postretirement plans and higher asset impairment charges.
- 8 -
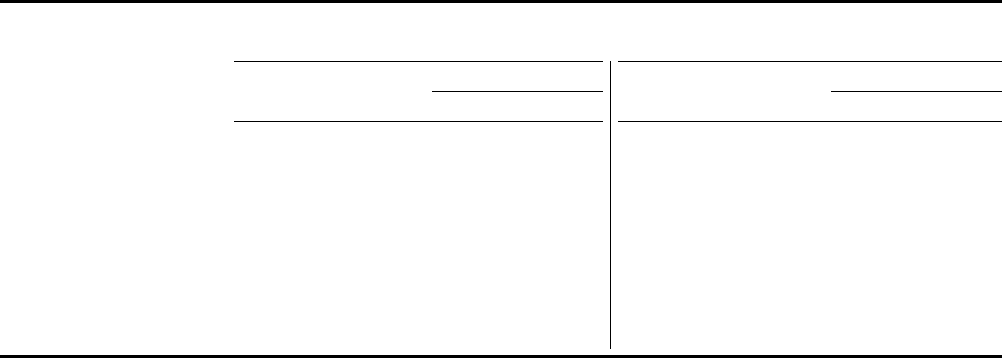
Pfizer’s effective tax rate on Reported income
(2)
for fourth-quarter 2022 decreased compared to the prior-year
quarter primarily due to a favorable change in the jurisdictional mix of earnings and global income tax
resolutions, partially offset by the non-recurrence of tax benefits associated with certain tax initiatives.
Adjusted
(3)
Income Statement Highlights
SELECTED ADJUSTED
(3)
COSTS AND EXPENSES
($ in millions) Fourth-Quarter Full-Year
2022 2021
% Change
2022 2021
% Change
Total Oper. Total Oper.
Adjusted
(3)
Cost of Sales $ 9,475 $ 9,710 (2%) 9% $ 34,096 $ 30,685 11% 20%
Percent of Revenues 39.0% 40.7% N/A N/A 34.0% 37.7% N/A N/A
Adjusted
(3)
SI&A Expenses 4,414 3,932 12% 17% 13,049 12,071 8% 11%
Adjusted
(3)
R&D Expenses 3,610 3,436 5% 7% 11,409 10,344 10% 12%
Adjusted
(3)
Other (Income)/
Deductions––net
($656) ($728) (10%) 2% ($1,954) ($2,475) (21%) (13%)
Effective Tax Rate on
Adjusted Income
(3)
11.1% 9.5% 11.7% 14.5 %
Reconciliations of certain Reported
(2)
to non-GAAP Adjusted
(3)
financial measures and associated footnotes can
be found in the financial tables section of this press release.
FULL-YEAR REVENUE SUMMARY (Full-Year 2022 vs. Full-Year 2021)
Full-year 2022 revenues totaled $100.3 billion, an increase of $19.0 billion, or 23%, compared to full-year 2021,
reflecting operational growth of $24.6 billion, or 30%, and an unfavorable impact of foreign exchange of $5.5
billion, or 7%. Excluding the revenue growth contributed by Paxlovid and Comirnaty
(1)
, revenues for the full-year
grew 2% operationally. Operational growth compared to the prior year was driven primarily by:
▪ Global sales of Paxlovid;
▪ Strong growth of Comirnaty
(1)
in developed markets;
▪ The launch of Prevnar 20 in the U.S. for the adult population;
▪ Continued strong growth of Eliquis globally;
▪ Vyndaqel family globally, partially offset by a planned price decrease in Japan; and
▪ Newly acquired products Nurtec ODT/Vydura and Oxbryta,
- 9 -
partially offset primarily by lower revenues for:
▪ Comirnaty
(1)
in emerging markets;
▪ Xeljanz, Chantix and Sutent globally; and
▪ Ibrance in developed Europe and the U.S.
RECENT NOTABLE DEVELOPMENTS (Since November 1, 2022)
Product Developments
▪ Comirnaty (COVID-19 vaccine, mRNA)
(9)
▪ Clinical and Research Developments
▪ In November 2022, Pfizer and BioNTech SE (BioNTech) announced updated clinical data from a
Phase 2/3 clinical trial demonstrating a robust neutralizing immune response one-month after a
30-µg booster dose of the companies’ Omicron BA.4/BA.5-adapted bivalent COVID-19 vaccine
(Pfizer-BioNTech COVID-19 Vaccine, Bivalent (Original and Omicron BA.4/BA.5)). Immune
responses against BA.4/BA.5 sublineages were substantially higher for those who received the
bivalent vaccine compared to the companies’ original COVID-19 vaccine, with a similar safety
and tolerability profile between both vaccines.
▪ In November 2022, Pfizer and BioNTech announced results from an analysis examining the
immune response induced by their Omicron BA.4/BA.5-adapted bivalent COVID-19 vaccine
against newer Omicron sublineages, including BA.4.6, BA.2.75.2, BQ.1.1 and XBB.1. These data
were posted on the preprint server bioRxiv and indicate that the companies’ bivalent vaccine
elicits a greater increase in neutralizing antibody titers than the companies’ original COVID-19
vaccine against these emerging Omicron sublineages.
▪ In November 2022, Pfizer and BioNTech announced that the companies have initiated a Phase 1
study to evaluate the safety, tolerability and immunogenicity of a next-generation COVID-19
vaccine candidate that aims to enhance SARS-CoV-2 T cell responses and potentially broaden
protection against COVID-19. This candidate, BNT162b4, is composed of a T cell antigen
mRNA encoding for SARS-CoV-2 non-spike proteins that are highly conserved across a broad
range of SARS-CoV-2 variants and will be evaluated in combination with the companies’
Omicron BA.4/BA.5-adapted bivalent COVID-19 vaccine.
▪ In December 2022, Pfizer and BioNTech announced the companies have received Fast Track
Designation from the U.S. Food and Drug Administration (FDA) for their mRNA-based
combination vaccine candidate for influenza and COVID-19, which aims to help prevent two
- 10 -
respiratory diseases with a single injection. The vaccine candidate is based on BioNTech’s
proprietary mRNA platform technology and contains mRNA strands encoding the wild-type
spike protein of SARS-CoV-2 and the spike protein of the Omicron sublineages BA.4/BA.5, as
well as mRNA strands encoding the hemagglutinin of four different influenza strains,
recommended for the Northern Hemisphere 2022/23 by the World Health Organization. A Phase
1 trial to examine the safety, tolerability, and immunogenicity of the combined influenza and
COVID-19 candidate vaccine among healthy adults was initiated in November 2022.
▪ Regulatory Developments
▪ In November 2022, Pfizer and BioNTech announced the European Medicines Agency (EMA)
Committee for Medicinal Products for Human Use (CHMP) recommended marketing
authorization for a 10-µg booster dose of the companies’ Omicron BA.4/BA.5-adapted bivalent
COVID-19 vaccine (Comirnaty Original/Omicron BA.4/BA.5 5-µg/5-µg) for children 5 through
11 years of age. The recommendation was subsequently endorsed by the European Commission
(EC).
▪ In December 2022, Pfizer and BioNTech announced the FDA granted Emergency Use
Authorization (EUA) of the companies’ Omicron BA.4/BA.5-adapted bivalent COVID-19
vaccine as the third 3-µg dose in the three-dose primary series for children 6 months through 4
years of age. Children in this age group can receive a primary series consisting of two 3-µg doses
of the original Pfizer-BioNTech COVID-19 vaccine followed by a third 3-µg dose of the bivalent
vaccine to complete the primary series.
▪ Ibrance (palbociclib) -- In December 2022, the FDA expanded the indication for Ibrance to include its use
in combination with an aromatase inhibitor (AI) for the treatment of HR+/HER2- metastatic breast cancer
(mBC), regardless of menopausal status. The approval expands on Ibrance’s existing indication for use in
combination with an AI as initial endocrine-based therapy in postmenopausal women or in men, and for use
with fulvestrant in patients with disease progression following endocrine therapy. Ibrance is now the only
CDK 4/6 inhibitor that is FDA-approved for the treatment of HR+/HER2- mBC in combination with either
an AI or fulvestrant regardless of menopausal status.
▪ Paxlovid (nirmatrelvir [PF-07321332] tablets and ritonavir tablets)
(9)
▪ In November 2022, Pfizer announced an agreement with the EC to supply Paxlovid to countries
participating in the Joint Procurement Agreement across Europe. This agreement is in addition to the
bilateral agreements Pfizer has previously signed with 17 EU Member States and will supply
participating countries up to 3.4 million treatment courses upon orders being placed. Pfizer began
delivery of the initial treatment quantities ordered by the participating countries in November.
▪ In December 2022, Pfizer announced it had reached an agreement with the U.S. Government for the
- 11 -

purchase of an additional 3.7 million treatment courses of Paxlovid. This purchase supplements the 20
million treatment courses previously contracted by and already delivered to the U.S. Government. The
additional 3.7 million treatment courses are planned for delivery in early 2023.
▪ In December 2022, Pfizer announced the FDA has extended the review period for the New Drug
Application (NDA) for Paxlovid. At the request of the FDA, Pfizer recently submitted additional
analyses of efficacy and safety data from the pivotal EPIC-HR (Evaluation of Protease Inhibition for
COVID-19 in High-Risk Patients) and supportive EPIC-SR (Evaluation of Protease Inhibition for
COVID-19 in Standard-Risk Patients) trials to be considered as part of its NDA for Paxlovid. Results
from these analyses are consistent with previously disclosed efficacy and safety data for the trials. In
order to allow time for a full review of the application, including the additional data analyses submitted,
the FDA has extended the Prescription Drug User Fee Act (PDUFA) goal date by three months to May
2023. Pfizer submitted its original NDA seeking approval of Paxlovid in June 2022 and was granted
priority review by the FDA.
▪ In January 2023, Pfizer announced that the CHMP of the EMA has recommended converting the
conditional Marketing Authorization for Paxlovid to standard (also referred to as “full”) Marketing
Authorization for the treatment of COVID-19 in adults who do not require supplemental oxygen and
who are at increased risk of the disease becoming severe. The EC will review the CHMP
recommendation and is soon expected to make a final decision.
▪ Prevnar 20 (20-valent pneumococcal conjugate vaccine) -- In January 2023, Pfizer announced that the
FDA accepted for review a supplemental Biologics License Application (sBLA) for its 20-valent
pneumococcal conjugate vaccine candidate for the prevention of invasive pneumococcal disease (IPD)
caused by the 20 Streptococcus pneumoniae serotypes contained in the vaccine in infants and children 6
weeks through 17 years of age, and for the prevention of otitis media caused by seven of the 20
Streptococcus pneumoniae serotypes contained in the vaccine. The PDUFA goal date for a decision by the
FDA is anticipated in April 2023.
Pipeline Developments
A comprehensive update of Pfizer’s development pipeline was published today and is now available at
www.pfizer.com/science/drug-product-pipeline. It includes an overview of Pfizer’s research and a list of
compounds in development with targeted indication and phase of development, as well as mechanism of action
for some candidates in Phase 1 and all candidates from Phase 2 through registration.
▪ Elranatamab (PF-06863135)
▪ In November 2022, Pfizer announced its investigational cancer immunotherapy, elranatamab, received
Breakthrough Therapy Designation from the FDA for the treatment of people with relapsed or
- 12 -
refractory multiple myeloma (RRMM). Elranatamab is a B-cell maturation antigen (BCMA)-CD3-
targeted bispecific antibody (BsAb).
▪ In December 2022, Pfizer announced 10.4 month follow-up data from the pivotal Phase 2
MagnetisMM-3 clinical trial suggesting elranatamab is efficacious and has a manageable safety profile
in patients with RRMM in a heavily pretreated population, who have received at least three classes of
prior therapies including a proteasome inhibitor, an immunomodulatory agent, and an anti-CD38
monoclonal antibody (i.e., triple-class refractory or exposed). These data were presented in an oral
session at the 64th American Society of Hematology Annual Meeting and Exposition 2022. Data from
the ongoing MagnetisMM-3 trial continue to be collected and will be shared as they mature.
▪ Etrasimod (Selective S1P Receptor Modulator) -- In December 2022, Pfizer announced that the FDA
accepted for review an NDA for etrasimod for individuals living with moderately-to-severely active
ulcerative colitis (UC). The FDA’s decision is expected in the second half of 2023. Pfizer also announced
that the EMA accepted the Marketing Authorization Application (MAA) for etrasimod in the same patient
population with a decision anticipated in the first half of 2024.
▪ Fidanacogene elaparvovec (Hemophilia B Gene Therapy) -- In December 2022, Pfizer announced
positive top-line results from the Phase 3 BENEGENE-2 study (NCT03861273) evaluating fidanacogene
elaparvovec for the treatment of adult males with moderately severe to severe hemophilia B. The
BENEGENE-2 study met its primary endpoint of non-inferiority and superiority in the annualized bleeding
rate of total bleeds post-fidanacogene elaparvovec infusion versus prophylaxis regimen with Factor IX,
administered as part of usual care. Fidanacogene elaparvovec was generally well-tolerated in the study, with
a safety profile consistent with Phase 1/2 results.
▪ PF-06886992 (Pentavalent (MenABCWY) Meningococcal Vaccine Candidate) -- In December 2022,
Pfizer announced the FDA accepted for review a Biologics License Application (BLA) for its investigational
pentavalent meningococcal vaccine candidate, MenABCWY. Pfizer submitted MenABCWY for the
prevention of meningococcal disease caused by the most common serogroups in individuals 10 through 25
years of age. If approved and recommended, the vaccine could help simplify the meningococcal vaccination
schedule and provide the broadest serogroup coverage of any meningococcal vaccine. The PDUFA goal date
for a decision by the FDA is in October 2023.
▪ RSVpreF (Respiratory Syncytial Virus (RSV) Bivalent Vaccine Candidate) -- In December 2022, Pfizer
announced that the FDA accepted for priority review a BLA for its RSV vaccine candidate, PF-06928316 or
RSVpreF, as submitted for the prevention of lower respiratory tract disease caused by RSV in individuals 60
years of age and older. The PDUFA goal date for a decision by the FDA is in May 2023.
▪ VLA15 (Lyme Disease Vaccine Candidate) -- In December 2022, Pfizer and Valneva SE (Valneva)
reported antibody persistence data six months after the completion of a three-dose (Month 0-2-6) or a two-
- 13 -
dose (Month 0-6) vaccination schedule with their Lyme disease vaccine candidate, VLA15, in both children
and adults. The data showed antibody levels declined over time, but remained above baseline six months
after completion of a three-dose (Month 0-2-6) or a two-dose (Month 0-6) vaccination schedule, with higher
antibody levels observed in the three-dose vaccination schedule versus the two-dose vaccination schedule.
No safety concerns were observed in the six-month observational follow-up. The three-dose vaccination
schedule is being used in the Phase 3 protocols for all participants.
Corporate Developments
▪ In December 2022, Pfizer business executives and scientific leadership provided updates on the company’s
potential near-term product launches, including investigational therapies and vaccines in migraine, RSV,
ulcerative colitis, alopecia, multiple myeloma and prostate cancer. Also discussed were key high-value
pipeline programs around sickle cell disease, dermatomyositis, hematological malignancies, obesity and type
2 diabetes, as well as Pfizer’s portfolio of mRNA vaccine candidates. If successful and approved, the
company anticipates these will be key drivers of Pfizer’s growth through 2030 and beyond.
▪ In January 2023, Pfizer announced a significant expansion of its commitment to An Accord for a Healthier
World (the Accord) by offering the full portfolio of medicines and vaccines for which it has global rights on
a not-for-profit basis to enable greater health for 1.2 billion people living in 45 lower-income countries. The
Accord, which was first launched in May 2022, originally included only patented products available in the
U.S. and EU, but now includes both patented and off-patent medicines and vaccines that treat or prevent
many of the greatest infectious and non-communicable disease threats faced today in lower-income
countries. As Pfizer launches new medicines and vaccines, those products will also be included in the
Accord portfolio on a not-for-profit basis.
- 14 -
For additional details, see the attached financial schedules, product revenue tables and disclosure notice.
(1) As used in this document, “Comirnaty” refers to, as applicable, and as authorized or approved, the Pfizer-
BioNTech COVID-19 Vaccine, the Pfizer-BioNTech COVID-19 Vaccine, Bivalent (Original and Omicron
BA.4/BA.5), the Comirnaty Original/Omicron BA.1 Vaccine, and Comirnaty Original/Omicron BA.4/
BA.5 Vaccine. “Comirnaty” includes direct sales and alliance revenues related to sales of the above-
mentioned vaccines, which are recorded within Pfizer’s Primary Care customer group. It does not include
revenues for certain Comirnaty-related manufacturing activities performed on behalf of BioNTech, which
are included in the Pfizer CentreOne contract development and manufacturing organization. Revenues
related to these manufacturing activities totaled $80 million and $188 million for the fourth-quarter and
full-year 2022, respectively, and $46 million and $320 million for the fourth-quarter and full-year 2021,
respectively.
(2) Revenues is defined as revenues in accordance with U.S. generally accepted accounting principles
(GAAP). Reported net income and its components are defined as net income attributable to Pfizer Inc. and
its components in accordance with U.S. GAAP. Reported diluted earnings per share (EPS) is defined as
diluted EPS attributable to Pfizer Inc. common shareholders in accordance with U.S. GAAP.
(3) Adjusted income and Adjusted diluted EPS are defined as U.S. GAAP net income attributable to Pfizer
Inc. common shareholders and Reported diluted EPS attributable to Pfizer Inc. common shareholders
before the impact of amortization of intangible assets, certain acquisition-related items, discontinued
operations and certain significant items. See the accompanying reconciliations of certain GAAP Reported
to Non-GAAP Adjusted information for fourth-quarter and full-year 2022 and 2021. Adjusted income and
its components and Adjusted diluted EPS measures are not, and should not be viewed as, substitutes for
U.S. GAAP net income and its components and diluted EPS
(2)
. See the Non-GAAP Financial Measure:
Adjusted Income sections of Management’s Discussion and Analysis of Financial Condition and Results of
Operations in Pfizer’s 2021 Annual Report on Form 10-K and Quarterly Report on Form 10-Q for the
quarterly period ended October 2, 2022 and the accompanying Non-GAAP Financial Measure: Adjusted
Income section of this press release for a definition of each component of Adjusted income as well as other
relevant information.
(4) Pfizer does not provide guidance for GAAP Reported financial measures (other than revenues and
acquired IPR&D expenses) or a reconciliation of forward-looking non-GAAP financial measures to the
most directly comparable GAAP Reported financial measures on a forward-looking basis because it is
unable to predict with reasonable certainty the ultimate outcome of unusual gains and losses, certain
acquisition-related expenses, gains and losses from equity securities, actuarial gains and losses from
pension and postretirement plan remeasurements, potential future asset impairments and pending litigation
- 15 -
without unreasonable effort. These items are uncertain, depend on various factors, and could have a
material impact on GAAP Reported results for the guidance period.
Financial guidance for full-year 2023 reflects the following:
▪ Does not assume the completion of any business development transactions not completed as of
December 31, 2022, except for signed transactions, if any, through mid-January 2023, which are
expected to give rise to acquired in-process R&D (IPR&D) expenses during fiscal 2023.
▪ Reflects an anticipated negative revenue impact of $0.3 billion due to recent and expected generic and
biosimilar competition for certain products that have recently lost patent protection or that are
anticipated to lose patent protection during fiscal-year 2023.
▪ Exchange rates assumed are as of mid-January 2023. Financial guidance reflects the anticipated
unfavorable impact of approximately $0.2 billion on revenues and approximately $0.02 on Adjusted
diluted EPS
(3)
as a result of changes in foreign exchange rates relative to the U.S. dollar compared to
foreign exchange rates from 2022.
▪ Guidance for Adjusted diluted EPS
(3)
assumes diluted weighted-average shares outstanding of
approximately 5.75 billion shares, and assumes no share repurchases in 2023.
(5) Pfizer’s fiscal year-end for international subsidiaries is November 30 while Pfizer’s fiscal year-end for
U.S. subsidiaries is December 31. Therefore, Pfizer’s fourth quarter and full year for U.S. subsidiaries
reflects the three and twelve months ended on December 31, 2022 and December 31, 2021, while Pfizer’s
fourth quarter and full year for subsidiaries operating outside the U.S. reflects the three and twelve months
ended on November 30, 2022 and November 30, 2021.
(6) Beginning in the third quarter of 2022, Pfizer made several organizational changes to further transform its
operations to better leverage its expertise in certain areas and in anticipation of potential future new
product or indication launches. Biopharma, Pfizer’s innovative science-based biopharmaceutical business,
is operating under a new commercial structure which is designed to better support and optimize
performance across three broad customer groups:
▪ Primary Care, consisting of the former Internal Medicine and Vaccines product portfolios, products
for COVID-19 prevention and treatment, and potential future mRNA and antiviral products.
▪ Specialty Care, consisting of the former Inflammation & Immunology, Rare Disease and Hospital
- 16 -
(excluding Paxlovid) product portfolios.
▪ Oncology, consisting of the former Oncology product portfolio.
(7) The following business development activity, among others, impacted financial results for the current or
prior fiscal year:
▪ On October 5, 2022, Pfizer announced the completion of its acquisition of Global Blood
Therapeutics, Inc. (GBT) for $68.50 per share in cash, for payments of approximately $5.3 billion, net
of cash acquired, plus repayment of third-party debt of $331 million for a total net cash deployment
of approximately $5.6 billion.
▪ On October 3, 2022, Pfizer announced the completion of its acquisition of all the outstanding shares
of Biohaven Pharmaceutical Holding Company Ltd. (Biohaven) not already owned by Pfizer for
$148.50 per share in cash, for payments of approximately $11.4 billion, net of cash acquired, plus
repayment of third-party debt of $863 million and redemption of Biohaven’s redeemable preferred
stock for $495 million, for a total net cash deployment of approximately $12.7 billion. Effective
immediately prior to the closing of the acquisition, Biohaven completed the spin-off of Biohaven Ltd.
(NYSE: BHVN), a new company that retained Biohaven’s non-calcitonin gene-related peptide
(CGRP) development stage pipeline compounds. Shares of Biohaven Ltd. were distributed to
Biohaven’s shareholders. Pfizer, a Biohaven shareholder, received a pro rata portion of the
company’s shares in the distribution and currently owns approximately 1.5% of Biohaven Ltd.
▪ On July 18, 2022, GlaxoSmithKline plc. (GSK) completed its demerger of the Consumer Healthcare
joint venture which became Haleon, an independent, publicly traded company listed on the London
Stock Exchange that holds the joint Consumer Healthcare business of GSK and Pfizer following the
demerger. For additional information, see Note 2C to the condensed consolidated financial statements
in Pfizer’s Quarterly Report on Form 10-Q for the quarterly period ended October 2, 2022.
▪ On June 9, 2022, Pfizer announced the completion of its acquisition of ReViral Ltd., a privately held,
clinical-stage biopharmaceutical company focused on discovering, developing and commercializing
novel antiviral therapeutics that target respiratory syncytial virus, for a total consideration of up to
$536 million, including upfront and development milestones. In connection with the closing of the
transaction, Pfizer recorded $426 million of acquired IPR&D expenses in its international third-
quarter 2022.
▪ On March 11, 2022, Pfizer announced the completion of its acquisition of Arena Pharmaceuticals,
Inc., a clinical-stage company developing innovative potential therapies for the treatment of several
- 17 -
immuno-inflammatory diseases, for $100 per share, in cash. The total fair value of the consideration
transferred was $6.6 billion ($6.2 billion, net of cash acquired), plus $138 million in payments to
Arena employees for previously unvested equity compensation awards recognized as an expense, for
a total net cash deployment of $6.4 billion.
▪ On December 31, 2021, Pfizer completed the sale of its Meridian subsidiary, the manufacturer of
EpiPen and other auto-injector products, which generated approximately $300 million in annual
revenues and which previously had been managed within the former Hospital therapeutic area.
Beginning in the fourth quarter of 2021, the financial results of Meridian are reflected as discontinued
operations for all periods presented.
▪ On December 24, 2021, Pfizer entered into a multi-year research collaboration with Beam
Therapeutics Inc. (Beam) to utilize Beam’s in vivo base editing programs, which use mRNA and lipid
nanoparticles, for three targets for rare genetic diseases of the liver, muscle and central nervous
system. Under the terms of the agreement, Pfizer paid Beam a $300 million upfront payment. If Pfizer
elects to opt in to licenses for all three targets, Beam would be eligible for up to an additional $1.05
billion in development, regulatory and commercial milestone payments for a potential total deal
consideration of up to $1.35 billion. Beam is also eligible to receive royalties on global net sales for
each licensed program.
▪ On November 17, 2021, Pfizer acquired all outstanding shares, warrants, options and deferred shares
not already owned by Pfizer of Trillium Therapeutics Inc., a clinical-stage immuno-oncology
company developing therapies targeting cancer immune evasion pathways and specific cell targeting
approaches, for a price of $18.50 per share in cash, for total consideration of $2.0 billion, net of cash
acquired. Pfizer accounted for the transaction as an asset acquisition since the lead asset, TTI-622,
represented substantially all of the fair value of the gross assets acquired. As a result, Pfizer recorded
a $2.1 billion charge in fourth-quarter 2021, representing the acquired in-process R&D asset.
▪ On November 9, 2021, Pfizer and Biohaven announced a strategic collaboration and license
agreement for Pfizer to commercialize rimegepant and zavegepant for the treatment and prevention of
migraines outside of the U.S., subject to regulatory approval. Upon the closing of the transaction on
January 4, 2022, Pfizer paid Biohaven $500 million, including an upfront payment of $150 million
and an equity investment of $350 million. Pfizer recognized $263 million for the upfront payment and
premium paid on its equity investment in acquired IPR&D expenses.
▪ On July 22, 2021, Arvinas Inc. (Arvinas) and Pfizer announced a global collaboration to develop and
commercialize ARV-471, an investigational oral PROTAC
®
(PROteolysis TArgeting Chimera)
estrogen receptor protein degrader. The estrogen receptor is a well-known disease driver in most
- 18 -

breast cancers. Under the terms of the agreement, Pfizer paid Arvinas $650 million upfront and made
a $350 million equity investment in Arvinas. Arvinas is also eligible to receive up to $400 million in
approval milestones and up to $1 billion in commercial milestones. The companies will equally share
worldwide development costs, commercialization expenses and profits.
(8) References to operational variances in this press release pertain to period-over-period changes that exclude
the impact of foreign exchange rates. Although exchange rate changes are part of Pfizer’s business, they
are not within Pfizer’s control and since they can mask positive or negative trends in the business, Pfizer
believes presenting operational variances excluding these foreign exchange changes provides useful
information to evaluate Pfizer’s results.
(9) Paxlovid and emergency uses of the Pfizer-BioNTech COVID-19 Vaccine or the Pfizer-BioNTech
COVID-19 Vaccine, Bivalent (Original and Omicron BA.4/BA.5), have not been approved or licensed by
the FDA. Paxlovid has not been approved, but has been authorized for emergency use by the FDA under
an EUA, for the treatment of mild-to-moderate COVID-19 in adults and pediatric patients (12 years of age
and older weighing at least 40 kg [88 lbs]) with positive results of direct SARS-CoV-2 viral testing, and
who are at high-risk for progression to severe COVID-19, including hospitalization or death. Emergency
uses of the Pfizer-BioNTech COVID-19 Vaccine and the Pfizer-BioNTech COVID-19 Vaccine, Bivalent
have been authorized by the FDA under an EUA to prevent COVID-19 in individuals aged 6 months and
older. The emergency uses are only authorized for the duration of the declaration that circumstances exist
justifying the authorization of emergency use of the medical product during the COVID-19 pandemic
under Section 564(b)(1) of the FFDCA unless the declaration is terminated or authorization revoked
sooner. Please see the EUA Fact Sheets at www.covid19oralrx.com and www.cvdvaccine-us.com.
Contacts: Media Investors
- 19 -

Fourth-Quarter % Incr. /
Full-Year
% Incr. /
2022 2021 (Decr.) 2022 2021 (Decr.)
Revenues
$ 24,290 $ 23,838 2
$ 100,330
$ 81,288 23
Costs and expenses:
Cost of sales
(2)
9,648 9,736 (1) 34,344 30,821 11
Selling, informational and administrative expenses
(2)
4,644 4,104 13 13,677 12,703 8
Research and development expenses
(2), (3)
3,615 3,445 5 11,428 10,360 10
Acquired in-process research and development expenses
(3)
73 2,469 (97) 953 3,469 (73)
Amortization of intangible assets
1,130 957 18 3,609 3,700 (2)
Restructuring charges and certain acquisition-related costs
(4)
795 135 * 1,375 802 71
Other (income)/deductions––net
(5)
(846) (835) 1 217 (4,878) *
Income from continuing operations before provision/(benefit)
for taxes on income
5,231 3,827 37 34,729 24,311 43
Provision/(benefit) for taxes on income
(6)
230 249 (8) 3,328 1,852 80
Income from continuing operations
5,000 3,578 40 31,401 22,459 40
Discontinued operations––net of tax
(1)
2 (187) * 6 (434) *
Net income before allocation to noncontrolling interests
5,002 3,391 48 31,407 22,025 43
Less: Net income attributable to noncontrolling interests
8 (2) * 35 45 (24)
Net income attributable to Pfizer Inc. common shareholders
$ 4,995 $ 3,393 47 $ 31,372 $ 21,979 43
Earnings per common share––basic:
Income from continuing operations attributable to Pfizer Inc.
common shareholders
$ 0.89 $ 0.64 39 $ 5.59 $ 4.00 40
Discontinued operations––net of tax
— (0.03) * — (0.08) *
Net income attributable to Pfizer Inc. common shareholders
$ 0.89 $ 0.60 47 $ 5.59 $ 3.92 43
Earnings per common share––diluted:
Income from continuing operations attributable to Pfizer Inc.
common shareholders
$ 0.87 $ 0.62 40 $ 5.47 $ 3.93 39
Discontinued operations––net of tax
— (0.03) * — (0.08) *
Net income attributable to Pfizer Inc. common shareholders
$ 0.87 $ 0.59 48 $ 5.47 $ 3.85 42
Weighted-average shares used to calculate earnings per
common share:
Basic
5,615 5,616 5,608 5,601
Diluted
5,743 5,768 5,733 5,708
* Indicates calculation not meaningful.
PFIZER INC. AND SUBSIDIARY COMPANIES
CONSOLIDATED STATEMENTS OF INCOME
(1)
(UNAUDITED)
(millions, except per share data)
- 20 -

(1) The financial statements present the three and twelve months ended December 31, 2022 and December 31, 2021.
Subsidiaries operating outside the U.S. are included for the three and twelve months ended November 30, 2022 and
November 30, 2021.
Business development activities completed in 2021 and 2022 impacted financial results in the periods presented.
Discontinued operations in 2022 relate to post-close adjustments and in 2021 relate to our former Meridian subsidiary
through December 31, 2021, the date of disposal, and post-close adjustments.
Certain amounts in the consolidated statements of income and associated notes may not add due to rounding. All
percentages have been calculated using unrounded amounts.
(2) Exclusive of amortization of intangible assets.
(3) In the first quarter of 2022, we began reporting Acquired in-process research and development expenses as a separate
line item in our consolidated statements of income. Acquired in-process research and development expenses includes
costs incurred in connection with (a) all upfront and milestone payments on collaboration and in-license agreements,
including premiums on equity securities and (b) asset acquisitions of acquired in-process research and development.
These costs were previously recorded in Research and development expenses. Prior periods have been revised to
conform to the current period presentation.
(4) Restructuring charges and certain acquisition-related costs include the following:
Fourth-Quarter Full-Year
(MILLIONS)
2022 2021 2022 2021
Restructuring charges/(credits)––acquisition-related costs
(a)
$ 64 $ — $ 138 $ (9)
Restructuring charges/(credits)––cost reduction initiatives
(b)
450 86 744 750
Restructuring charges/(credits) 514 86 882 741
Transaction costs
(c)
102 20 144 20
Integration costs and other
(d)
178 30 348 41
Restructuring charges and certain acquisition-related costs $ 795 $ 135 $ 1,375 $ 802
(a) Includes employee termination costs, asset impairments and other exit costs associated with business
combinations.
(b) Includes employee termination costs, asset impairments and other exit costs not associated with acquisitions.
(c) Transaction costs represent external costs for banking, legal, accounting and other similar services.
(d) Integration costs and other represent external, incremental costs directly related to integrating acquired businesses,
such as expenditures for consulting and the integration of systems and processes, and certain other qualifying
costs.
(5) Components of Other (income)/deductions––net include:
Fourth-Quarter Full-Year
(MILLIONS)
2022 2021 2022 2021
Interest income $ (137)
$ (14)
$ (251)
$ (36)
Interest expense
313 315 1,238 1,291
Net interest expense
176 301 987 1,255
Royalty-related income
(217) (208) (845) (857)
Net (gains)/losses on asset disposals
(6) — — (99)
Net (gains)/losses recognized during the period on equity securities
(79) 257 1,273 (1,344)
Income from collaborations, out-licensing arrangements and sales
of compound/product rights
(170) (79) (188) (396)
Net periodic benefit costs/(credits) other than service costs
(555) (913) (849) (2,547)
Certain legal matters, net
55 69 230 182
Certain asset impairments
(a)
221 86 421 86
Haleon/Consumer Healthcare JV equity method (income)/loss
(154) (163) (436) (471)
Other, net
(118) (185) (378) (687)
Other (income)/deductions––net $ (846) $ (835) $ 217 $ (4,878)
(a) The amounts in the fourth quarter of 2022 represent, and for full-year 2022 include, intangible asset impairment
charges of $221 million, primarily related to developed technology rights acquired in connection with our
Hospira, Inc. acquisition. The amount for full-year 2022 also includes an intangible asset impairment charge of
PFIZER INC. AND SUBSIDIARY COMPANIES
NOTES TO CONSOLIDATED STATEMENTS OF INCOME - (UNAUDITED)
- 21 -
$200 million associated with the discontinuation of the PF-07265803 (lamin A/C protein (LMNA)-related dilated
cardiomyopathy) clinical program.
(6) Our effective tax rates for income from continuing operations were: 4.4% and 9.6% in the three and twelve months
ended December 31, 2022, respectively, and 6.5% and 7.6% in the three and twelve months ended December 31, 2021,
respectively. The effective tax rate for the fourth quarter of 2022 compared to the fourth quarter of 2021 decreased due
to a favorable change in the jurisdictional mix of earnings and global income tax resolutions, partially offset by the
non-recurrence of tax benefits associated with certain tax initiatives. The higher effective tax rate for full-year 2022,
compared to full-year 2021, was mainly due to the non-recurrence of certain initiatives executed in 2021 associated
with our investment in the Consumer Healthcare joint venture with GlaxoSmithKline plc, partially offset by tax
benefits in 2022 related to global income tax resolutions in multiple tax jurisdictions spanning multiple tax years that
included the closing of U.S. Internal Revenue Service audits covering five tax years.
PFIZER INC. AND SUBSIDIARY COMPANIES
NOTES TO CONSOLIDATED STATEMENTS OF INCOME - (UNAUDITED)
- 22 -

Adjusted income is an alternative measure of performance used by management to evaluate our overall performance as a
supplement to our GAAP Reported performance measures. As such, we believe that investors’ understanding of our
performance is enhanced by disclosing this measure. We use Adjusted income, certain components of Adjusted income and
Adjusted diluted EPS to present the results of our major operations––the discovery, development, manufacture, marketing,
sale and distribution of biopharmaceutical products worldwide––prior to considering certain income statement elements as
follows:
Measure Definition
Relevance of Metrics to Our
Business Performance
Adjusted income
Net income attributable to Pfizer Inc. common shareholders
(a)
before the impact of amortization of intangible assets, certain
acquisition-related items, discontinued operations and certain
significant items
• Provides investors useful
information to:
◦ evaluate the normal
recurring operational
activities, and their
components, on a
comparable year-over-year
basis
◦ assist in modeling expected
future performance on a
normalized basis
• Provides investors insight
into the way we manage our
budgeting and forecasting,
how we evaluate and manage
our recurring operations and
how we reward and
compensate our senior
management
(b)
Adjusted cost of sales, Adjusted
selling, informational and
administrative expenses, Adjusted
research and development expenses
and Adjusted other (income)/
deductions––net
Cost of sales, Selling, informational and administrative
expenses, Research and development expenses and Other
(income)/deductions––net
(a)
, each before the impact of
amortization of intangible assets, certain acquisition-related
items, discontinued operations and certain significant items,
which are components of the Adjusted income measure
Adjusted diluted EPS
EPS attributable to Pfizer Inc. common shareholders––diluted
(a)
before the impact of amortization of intangible assets, certain
acquisition-related items, discontinued operations and certain
significant items
(a)
Most directly comparable GAAP measure.
(b)
Beginning in the first quarter of 2022, we no longer exclude any expenses for acquired IPR&D from our non-GAAP Adjusted results but we continue to
exclude certain of these expenses for our financial results for annual incentive compensation purposes.
Adjusted income and its components and Adjusted diluted EPS are non-GAAP financial measures that have no standardized
meaning prescribed by GAAP and, therefore, are limited in their usefulness to investors. Because of their non-standardized
definitions, they may not be comparable to the calculation of similar measures of other companies and are presented to permit
investors to more fully understand how management assesses performance. A limitation of these measures is that they
provide a view of our operations without including all events during a period, and do not provide a comparable view of our
performance to peers. These measures are not, and should not be viewed as, substitutes for their most directly comparable
GAAP measures of Net income attributable to Pfizer Inc. common shareholders, components of Net income attributable to
Pfizer Inc. common shareholders and EPS attributable to Pfizer Inc. common shareholders—diluted, respectively.
We also recognize that, as internal measures of performance, these measures have limitations, and we do not restrict our
performance-management process solely to these measures. We also use other tools designed to achieve the highest levels of
performance. For example, our R&D organization has productivity targets, upon which its effectiveness is measured. In
addition, total shareholder return, both on an absolute basis and relative to a publicly traded pharmaceutical index, plays a
significant role in determining payouts under certain of our incentive compensation plans.
Beginning in the first quarter of 2022, our reconciliation of certain GAAP Reported to non-GAAP Adjusted information is
updated to reflect the following, and prior period information has been revised to conform to the current period presentation:
Adjusted Income and Adjusted Diluted EPS
Acquired IPR&D—Non-GAAP Adjusted financial measures include expenses for all acquired in-process research and
development (IPR&D) costs incurred in connection with upfront and milestone payments on collaboration and in-license
agreements, including premiums on equity securities, as well as asset acquisitions of acquired IPR&D. Previously, certain of
these items were excluded from our non-GAAP Adjusted results. Acquired IPR&D expenses that previously would have
been excluded from non-GAAP Adjusted income but are now included in both GAAP Reported income and non-GAAP
Adjusted income had no impact in the fourth quarter of 2022 and were approximately: (i) $765 million pre-tax ($665 million,
net of tax), or $0.12 per share, in full-year 2022; (ii) $2.4 billion pre-tax ($1.8 billion, net of tax), or $0.32 per share, in the
fourth quarter of 2021; and (iii) $3.3 billion pre-tax ($2.6 billion, net of tax), or $0.45 per share, in full-year 2021.
PFIZER INC. AND SUBSIDIARY COMPANIES
NON-GAAP FINANCIAL MEASURE: ADJUSTED INCOME
- 23 -

Amortization of Intangible Assets—We began excluding all amortization of intangibles from non-GAAP Adjusted income,
compared to excluding only amortization of intangibles related to large mergers or acquisitions under the prior methodology,
and presenting it as a separate reconciling line. Previously, the adjustment under the prior methodology was included as part
of a reconciling line entitled “Purchase accounting adjustments” that we no longer separately present. The impact of this
policy change resulted in benefits of $0.02 and $0.06 on Adjusted diluted EPS in the fourth quarter and full-year 2022,
respectively, and $0.02 and $0.09 in the fourth quarter and full-year 2021, respectively.
Acquisition-Related Items—Acquisition-related items may now include purchase accounting impacts that previously would
have been included as part of a reconciling line entitled “Purchase accounting adjustments” that we no longer separately
present, such as: (i) the incremental charge to cost of sales from the sale of acquired inventory that was written up to fair
value, (ii) depreciation related to the increase/decrease in fair value of acquired fixed assets, (iii) amortization related to the
increase in fair value of acquired debt and (iv) the fair value changes for contingent consideration.
See the reconciliations of certain GAAP Reported to Non-GAAP Adjusted information for the fourth quarter and full-year
2022 and 2021 below and the Non-GAAP Financial Measure: Adjusted Income sections of Management’s Discussion and
Analysis of Financial Condition and Results of Operations in Pfizer’s 2021 Annual Report on Form 10-K and Quarterly
Report on Form 10-Q for the quarterly period ended October 2, 2022 for additional information.
PFIZER INC. AND SUBSIDIARY COMPANIES
NON-GAAP FINANCIAL MEASURE: ADJUSTED INCOME
- 24 -
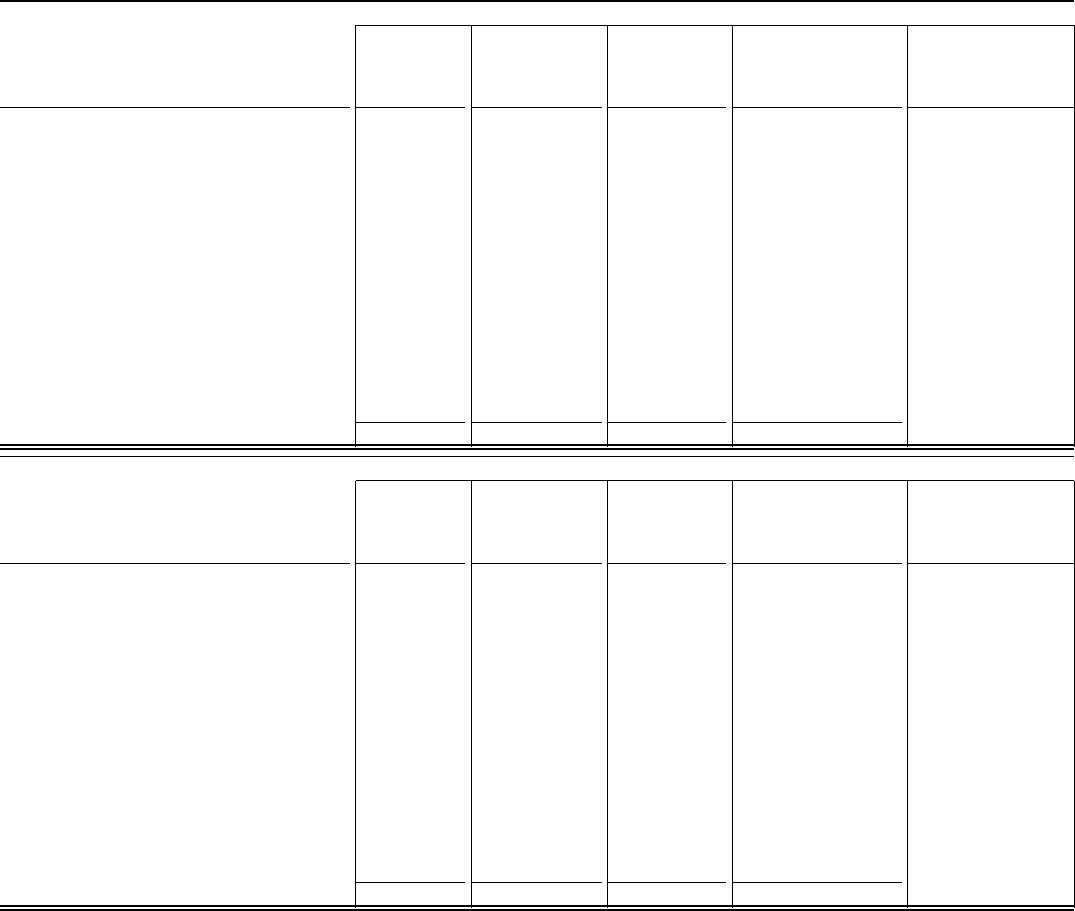
Fourth-Quarter 2022
Data presented will not (in all cases) aggregate to
totals. Cost of sales
(1)
Selling,
informational and
administrative
expenses
(1)
Other (income)/
deductions––
net
(1)
Net income attributable
to Pfizer Inc. common
shareholders
(1), (2)
Earnings per common
share attributable to
Pfizer Inc. common
shareholders––diluted
GAAP Reported
$ 9,648 $ 4,644 $ (846) $ 4,995 $ 0.87
Amortization of intangible assets
— — — 1,130
Acquisition-related items
(131) (2) (23) 501
Discontinued operations
(3)
— — — (12)
Certain significant items:
Restructuring charges/(credits) and implementation
costs and additional depreciation—asset
restructuring
(4)
(26) (217) — 695
Certain asset impairments
(5)
— — (221) 221
(Gains)/losses on equity securities
— — 78 (78)
Actuarial valuation and other pension and
postretirement plan (gains)/losses
— — 455 (455)
Other
(6)
(16) (11) (100) 130
Income tax provision—non-GAAP items
(576)
Non-GAAP Adjusted
$ 9,475 $ 4,414 $ (656)
(7)
$ 6,551 $ 1.14
Full-Year Ended December 31, 2022
Data presented will not (in all cases) aggregate to
totals. Cost of sales
(1)
Selling,
informational and
administrative
expenses
(1)
Other (income)/
deductions––
net
(1)
Net income attributable
to Pfizer Inc. common
shareholders
(1), (2)
Earnings per common
share attributable to
Pfizer Inc. common
shareholders––diluted
GAAP Reported
$ 34,344 $ 13,677 $ 217 $ 31,372 $ 5.47
Amortization of intangible assets
— — — 3,609
Acquisition-related items
(119) (7) (74) 832
Discontinued operations
(3)
— — — (21)
Certain significant items:
Restructuring charges/(credits) and implementation
costs and additional depreciation—asset
restructuring
(4)
(88) (562) — 1,396
Certain asset impairments
(5)
— — (421) 421
(Gains)/losses on equity securities
— — (1,270) 1,270
Actuarial valuation and other pension and
postretirement plan (gains)/losses
— — 230 (230)
Other
(6)
(40) (59) (636) 752
Income tax provision—Non-GAAP items
(1,683)
Non-GAAP Adjusted
$ 34,096 $ 13,049 $ (1,954)
(7)
$ 37,717 $ 6.58
See end of tables for notes.
PFIZER INC. AND SUBSIDIARY COMPANIES
RECONCILIATIONS OF GAAP REPORTED TO NON-GAAP ADJUSTED INFORMATION
CERTAIN LINE ITEMS - (UNAUDITED)
(millions, except per share data)
- 25 -
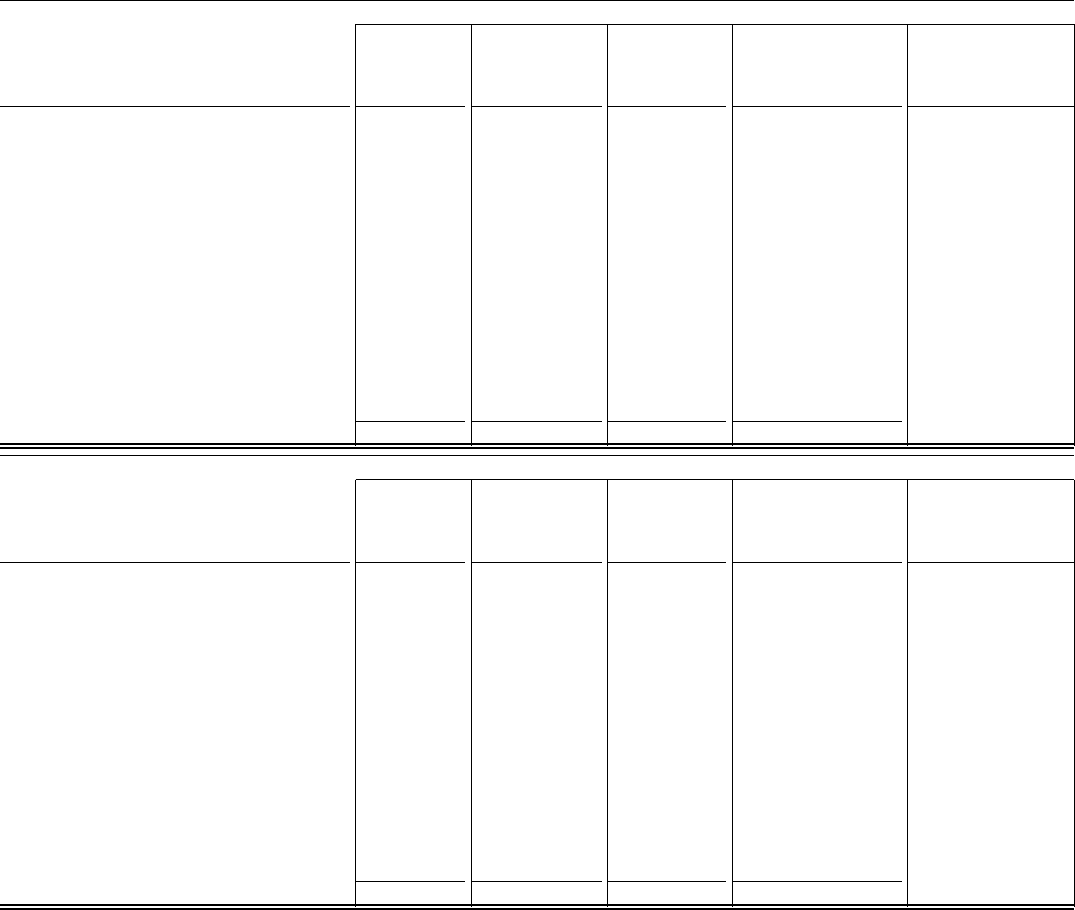
Fourth-Quarter 2021
Data presented will not (in all cases) aggregate to
totals. Cost of sales
(1)
Selling,
informational and
administrative
expenses
(1)
Other (income)/
deductions––
net
(1)
Net income attributable
to Pfizer Inc. common
shareholders
(1), (2)
Earnings per common
share attributable to
Pfizer Inc. common
shareholders––diluted
GAAP Reported
$ 9,736 $ 4,104 $ (835) $ 3,393 $ 0.59
Amortization of intangible assets
— (9) (1) 968
Acquisition-related items
7 (1) (83) 125
Discontinued operations
(3)
— — — 232
Certain significant items:
Restructuring charges/(credits) and implementation
costs and additional depreciation—asset
restructuring
(4)
(26) (140) — 252
Certain asset impairments
— — (86) 86
(Gains)/losses on equity securities
— — (259) 259
Actuarial valuation and other pension and
postretirement plan (gains)/losses
— — 669 (669)
Other
(6)
(7) (22) (134) 172
Income tax provision—non-GAAP items
(274)
Non-GAAP Adjusted
$ 9,710 $ 3,932 $ (728)
(7)
$ 4,543 $ 0.79
Full-Year Ended December 31, 2021
Data presented will not (in all cases) aggregate to
totals. Cost of sales
(1)
Selling,
informational and
administrative
expenses
(1)
Other (income)/
deductions––
net
(1)
Net income attributable
to Pfizer Inc. common
shareholders
(1), (2)
Earnings per common
share attributable to
Pfizer Inc. common
shareholders––diluted
GAAP Reported
$ 30,821 $ 12,703 $ (4,878) $ 21,979 $ 3.85
Amortization of intangible assets
— (38) (2) 3,746
Acquisition-related items
25 (3) (114) 139
Discontinued operations
(3)
— — — 585
Certain significant items:
Restructuring charges/(credits) and implementation
costs and additional depreciation—asset
restructuring
(4)
(108) (450) — 1,309
Certain asset impairments
— — (86) 86
(Gains)/losses on equity securities
— — 1,338 (1,338)
Actuarial valuation and other pension and
postretirement plan (gains)/losses
— — 1,601 (1,601)
Other
(6)
(52) (141) (334) 542
Income tax provision—Non-GAAP items
(2,250)
Non-GAAP Adjusted
$ 30,685 $ 12,071 $ (2,475)
(7)
$ 23,196 $ 4.06
PFIZER INC. AND SUBSIDIARY COMPANIES
RECONCILIATIONS OF GAAP REPORTED TO NON-GAAP ADJUSTED INFORMATION
CERTAIN LINE ITEMS - (UNAUDITED)
(millions, except per share data)
- 26 -

(1) Items that reconcile GAAP Reported to non-GAAP Adjusted balances are shown pre-tax. Our effective tax rates for
GAAP Reported income from continuing operations were: 4.4% and 9.6% in the three and twelve months ended
December 31, 2022, respectively, and 6.5% and 7.6% in the three and twelve months ended December 31, 2021,
respectively. See Note (6) to the Consolidated Statements of Income above. Our effective tax rates for non-GAAP
Adjusted income were: 11.1% and 11.7% in the three and twelve months ended December 31, 2022, respectively, and
9.5% and 14.5% in the three and twelve months ended December 31, 2021, respectively.
(2) The amounts for fourth quarter and full-year 2022 and 2021 include reconciling amounts for Research and
development expenses that are not material.
(3) For 2022, relates to post-close adjustments and for 2021, relates to our former Meridian subsidiary through December
31, 2021, the date of disposal, and post-close adjustments.
(4) Includes employee termination costs, asset impairments and other exit costs related to our cost-reduction and
productivity initiatives not associated with acquisitions.
(5) See Note (5) to the Consolidated Statements of Income above.
(6) For 2022, the total Other (income)/deductions––net adjustments of $100 million in the fourth quarter and $636 million
for the full-year primarily include (i) charges of $34 million and $307 million, respectively, mostly representing our
equity-method accounting pro rata share of restructuring charges and costs of preparing for separation from
GlaxoSmithKline plc (GSK) recorded by Haleon/the Consumer Healthcare joint venture (JV), and adjustments to our
equity-method basis differences which are also related to the separation of Haleon/the Consumer Healthcare JV from
GSK, and (ii) charges for certain legal matters of $55 million and $230 million, respectively. For 2021, the total Other
(income)/deductions––net adjustments of $134 million in the fourth quarter and $334 million for the full-year
primarily included (i) charges of $69 million and $162 million, respectively, for certain legal matters and (ii) charges
of $50 million and $185 million mostly representing our equity-method accounting pro rata share of restructuring
charges and costs of preparing for separation from GSK recorded by the Consumer Healthcare JV. For full-year 2021,
Selling, informational and administrative expenses of $141 million primarily included costs for consulting, legal, tax
and advisory services associated with a non-recurring internal reorganization of legal entities.
(7) The components of non-GAAP Adjusted Other (income)/deductions––net include the following:
Fourth-Quarter Full-Year
(MILLIONS
2022 2021 2022 2021
Interest income
$ (137) $ (14) $ (251) $ (36)
Interest expense
315 317 1,247 1,297
Net interest expense
178 303 995 1,262
Royalty-related income
(217) (208) (845) (857)
Net (gains)/losses on asset disposals
(6) — (7) (42)
Net (gains)/losses recognized during the period on equity securities
(1) (2) 4 (6)
Income from collaborations, out-licensing arrangements and sales of
compound/product rights
(170) (79) (188) (396)
Net periodic benefit costs/(credits) other than service costs
(99) (244) (619) (946)
Certain legal matters, net
— — — 20
Haleon/Consumer Healthcare JV equity method (income)/loss
(188) (213) (743) (656)
Other, net
(153) (286) (552) (854)
Non-GAAP Adjusted Other (income)/deductions––net $ (656) $ (728) $ (1,954) $ (2,475)
See Note (5) to the Consolidated Statements of Income above for additional information on the components
comprising GAAP Reported Other (income)/deductions––net.
PFIZER INC. AND SUBSIDIARY COMPANIES
NOTES TO RECONCILIATIONS OF GAAP REPORTED TO NON-GAAP ADJUSTED INFORMATION
CERTAIN LINE ITEMS - (UNAUDITED)
- 27 -

WORLDWIDE UNITED STATES
TOTAL INTERNATIONAL
(a)
2022 2021
% Change
2022 2021
% Change
2022 2021
% Change
(MILLIONS)
Total Oper. Total Total Oper.
TOTAL REVENUES $ 24,290 $ 23,838 2% 13% $ 8,481 $ 7,680 10% $ 15,808 $ 16,159 (2%) 14%
GLOBAL BIOPHARMACEUTICALS BUSINESS (BIOPHARMA)
(b)
$ 23,922 $ 23,456 2% 13% $ 8,382 $ 7,564 11% $ 15,539 $ 15,891 (2%) 14%
Primary Care $ 17,348 $ 16,225 7% 20% $ 4,815 $ 3,815 26% $ 12,533 $ 12,410 1% 18%
Comirnaty direct sales and alliance revenues 11,329 12,504 (9%) 3% 2,472 2,151 15% 8,857 10,353 (14%) 1%
Paxlovid 1,834 76 * * — 76 * 1,834 — * *
Eliquis alliance revenues and direct sales 1,479 1,500 (1%) 5% 843 720 17% 636 780 (18%) (7%)
Prevnar family
(c)
1,736 1,302 33% 40% 1,022 571 79% 714 731 (2%) 10%
Premarin family 128 143 (11%) (10%) 119 135 (12%) 9 8 11% 23%
BMP2 76 80 (5%) (5%) 76 80 (5%) — — — —
Nimenrix 48 48 (1%) 14% — — — 48 48 (1%) 14%
Nurtec ODT/Vydura 211 — * * 211 — * 1 — * *
FSME-IMMUN/TicoVac 23 24 (6%) 9% 1 — * 22 24 (9%) 5%
Toviaz 16 64 (74%) (67%) (4) 22 * 21 42 (51%) (39%)
Trumenba 15 16 (11%) (9%) 12 14 (14%) 3 3 1% 16%
Chantix/Champix — (11) * * — 1 * — (12) * *
All other Primary Care 452 478 (5%) 5% 65 45 43% 388 433 (10%) 1%
Specialty Care $ 3,566 $ 3,989 (11%) (3%) $ 1,553 $ 1,700 (9%) $ 2,013 $ 2,289 (12%) 1%
Vyndaqel family
(d)
680 561 21% 31% 355 251 41% 326 310 5% 22%
Xeljanz 493 721 (32%) (28%) 327 516 (37%) 165 206 (20%) (7%)
Enbrel (Outside the U.S. and Canada) 236 297 (21%) (8%) — — — 236 297 (21%) (8%)
Sulperazon 189 169 12% 24% — — — 189 169 12% 24%
Inflectra 129 171 (25%) (20%) 62 108 (43%) 68 63 7% 19%
Ig Portfolio
(e)
135 119 14% 14% 135 119 14% — — — —
BeneFIX 101 109 (8%) 1% 54 56 (4%) 46 53 (12%) 6%
Zavicefta 109 107 2% 15% — — — 109 107 2% 15%
Genotropin 99 106 (6%) 8% 27 25 7% 72 80 (10%) 8%
Zithromax 81 81 — 13% 1 1 11% 80 80 — 13%
Medrol 93 112 (17%) (11%) 43 39 9% 50 72 (31%) (22%)
Fragmin 67 82 (19%) (9%) 1 1 23% 65 81 (19%) (9%)
Somavert 66 74 (11%) (3%) 27 28 (1%) 39 47 (17%) (5%)
Refacto AF/Xyntha 52 69 (25%) (14%) 11 13 (14%) 40 56 (28%) (14%)
Vfend 55 63 (13%) (1%) 1 3 (60%) 54 60 (11%) 2%
Oxbryta 73 — * * 72 — * — — — —
All other Anti-infectives 348 451 (23%) (15%) 109 139 (22%) 240 312 (23%) (12%)
All other Specialty Care 561 696 (19%) (15%) 327 399 (18%) 234 297 (21%) (11%)
Oncology $ 3,007 $ 3,242 (7%) (3%) $ 2,014 $ 2,050 (2%) $ 993 $ 1,192 (17%) (4%)
Ibrance 1,279 1,398 (8%) (4%) 876 879 — 403 519 (22%) (10%)
Xtandi alliance revenues 320 306 5% 5% 320 306 5% — — — —
Inlyta 243 260 (7%) (2%) 164 152 8% 79 109 (28%) (17%)
Bosulif 150 145 3% 9% 98 94 4% 52 51 2% 18%
Zirabev 130 133 (2%) 3% 97 86 13% 33 47 (29%) (16%)
Xalkori 103 122 (16%) (7%) 27 25 7% 76 97 (22%) (11%)
Ruxience 101 148 (32%) (30%) 83 135 (39%) 18 13 43% 59%
Retacrit 86 122 (30%) (27%) 65 95 (32%) 21 26 (21%) (9%)
Sutent 60 137 (56%) (50%) 4 13 (69%) 56 124 (54%) (48%)
Lorbrena 95 73 30% 41% 48 38 26% 47 35 35% 58%
Bavencio alliance revenues 72 56 30% 44% 27 23 15% 46 33 40% 64%
Aromasin 61 53 16% 29% 1 1 (29%) 61 52 17% 30%
Besponsa 55 47 16% 24% 31 26 19% 24 21 12% 29%
Trazimera 54 66 (19%) (15%) 21 38 (46%) 33 28 19% 27%
Braftovi 37 51 (26%) (26%) 36 51 (29%) 1 — * *
Mektovi 46 42 9% 9% 46 43 8% — — — —
All other Oncology 113 83 37% 43% 70 45 58% 43 39 12% 26%
PFIZER CENTREONE
(b)
$ 368 $ 382 (4%) 1% $ 99 $ 115 (14%) $ 269 $ 267 1% 7%
Total Alliance revenues included above $ 2,217 $ 1,934 15% 19% $ 1,216 $ 1,061 15% $ 1,001 $ 873 15% 24%
See end of tables for notes.
PFIZER INC. - REVENUES
FOURTH-QUARTER 2022 and 2021 - (UNAUDITED)
- 28 -

DEVELOPED EUROPE
(f)
DEVELOPED REST OF WORLD
(g)
EMERGING MARKETS
(h)
2022 2021
% Change
2022 2021
% Change
2022 2021
% Change
(MILLIONS)
Total Oper. Total Oper. Total Oper.
TOTAL INTERNATIONAL REVENUES $ 7,277 $ 4,499 62% 86% $ 5,107 $ 3,889 31% 59% $ 3,424 $ 7,771 (56%) (51%)
GLOBAL BIOPHARMACEUTICALS BUSINESS (BIOPHARMA)
(b)
$ 7,064 $ 4,354 62% 87% $ 5,084 $ 3,863 32% 60% $ 3,390 $ 7,674 (56%) (51%)
Primary Care $ 6,008 $ 3,068 96% * $ 4,486 $ 3,088 45% 77% $ 2,040 $ 6,254 (67%) (64%)
Comirnaty direct sales and alliance revenues 4,285 2,177 97% * 3,642 2,767 32% 62% 930 5,409 (83%) (81%)
Paxlovid 995 — * * 599 — * * 241 — * *
Eliquis alliance revenues and direct sales 359 423 (15%) (1%) 82 119 (31%) (15%) 194 238 (19%) (13%)
Prevnar family
(c)
174 223 (22%) (9%) 86 102 (16%) — 455 405 12% 22%
Premarin family — — — — 5 5 — 12% 3 2 39% 51%
BMP2 — — — — — — — — — — — —
Nimenrix 24 30 (22%) (9%) 5 2 * * 19 16 18% 35%
Nurtec ODT/Vydura — — — — — — — — 1 — * *
FSME-IMMUN/TicoVac 20 22 (8%) 7% — — — — 2 2 (11%) 2%
Toviaz 5 18 (71%) (67%) 14 21 (35%) (19%) 2 3 (38%) (10%)
Trumenba 2 3 (20%) (7%) — — — — 1 — * *
Chantix/Champix — (1) * * — (1) * * — (10) * *
All other Primary Care 143 173 (17%) (7%) 52 72 (28%) (14%) 193 188 3% 15%
Specialty Care $ 670 $ 729 (8%) 6% $ 390 $ 535 (27%) (14%) $ 954 $ 1,026 (7%) 4%
Vyndaqel family
(d)
239 155 55% 78% 62 142 (56%) (46%) 24 13 89% *
Xeljanz 62 73 (15%) (1%) 51 71 (29%) (15%) 52 61 (15%) (4%)
Enbrel (Outside the U.S. and Canada) 87 127 (32%) (21%) 42 60 (30%) (13%) 107 110 (3%) 9%
Sulperazon — — — — 1 2 (53%) (40%) 188 167 13% 24%
Inflectra 31 40 (22%) (10%) 34 21 65% 78% 3 3 (11%) 2%
Ig Portfolio
(e)
— — — — — — — — — — — —
BeneFIX 11 16 (29%) (17%) 12 15 (16%) — 23 22 3% 26%
Zavicefta 27 31 (11%) 3% — — — — 82 76 7% 20%
Genotropin 25 30 (15%) (1%) 20 25 (19%) — 27 26 5% 27%
Zithromax 15 16 (4%) 12% 4 5 (23%) (4%) 60 58 3% 14%
Medrol 14 16 (16%) (2%) 10 11 (15%) (1%) 27 45 (40%) (34%)
Fragmin 35 42 (16%) (2%) 14 15 (9%) (2%) 16 24 (32%) (27%)
Somavert 29 35 (17%) (4%) 5 7 (30%) (21%) 5 5 (1%) 12%
Refacto AF/Xyntha 15 27 (46%) (37%) 3 6 (42%) (34%) 23 23 (3%) 18%
Vfend 3 4 (37%) (26%) 10 11 (10%) 13% 41 45 (9%) 2%
Oxbryta — — — — — — — — — — — —
All other Anti-infectives 63 79 (20%) (8%) 23 30 (21%) (5%) 154 204 (25%) (15%)
All other Specialty Care 14 39 (64%) (59%) 99 115 (14%) (3%) 122 143 (15%) (5%)
Oncology $ 387 $ 557 (31%) (19%) $ 209 $ 241 (13%) 5% $ 397 $ 394 1% 11%
Ibrance 188 261 (28%) (16%) 98 116 (16%) 1% 118 142 (17%) (7%)
Xtandi alliance revenues — — — — — — — — — — — —
Inlyta 21 51 (59%) (51%) 16 23 (30%) (15%) 42 35 19% 29%
Bosulif 23 24 (5%) 10% 16 17 (7%) 16% 13 10 35% 42%
Zirabev 22 34 (36%) (26%) 9 11 (11%) 7% 2 2 8% 27%
Xalkori 19 23 (15%) (2%) 9 11 (18%) (1%) 47 63 (25%) (16%)
Ruxience 6 6 (10%) 6% 6 5 20% 35% 6 1 * *
Retacrit 20 25 (23%) (10%) — — — — 1 1 16% 21%
Sutent 9 48 (81%) (78%) 12 18 (31%) (17%) 35 58 (40%) (33%)
Lorbrena 17 16 6% 23% 11 10 5% 29% 19 9 * *
Bavencio alliance revenues 22 17 29% 50% 15 13 19% 50% 9 3 * *
Aromasin 5 7 (22%) (10%) 1 2 (22%) (3%) 54 43 24% 38%
Besponsa 9 8 6% 23% 6 8 (31%) (15%) 10 5 93% *
Trazimera 9 12 (21%) (9%) 2 2 (4%) 12% 22 14 57% 60%
Braftovi — — — — 1 — * * — — — —
Mektovi — — — — — — — — — — — —
All other Oncology 17 25 (31%) (21%) 6 4 41% 57% 20 9 * *
PFIZER CENTREONE
(b)
$ 213 $ 145 47% 54% $ 23 $ 25 (11%) 10% $ 34 $ 97 (65%) (65%)
Total Alliance revenues included above $ 887 $ 584 52% 62% $ 101 $ 138 (26%) (10%) $ 13 $ 152 (91%) (90%)
PFIZER INC.
INTERNATIONAL REVENUES BY GEOGRAPHIC REGION
FOURTH-QUARTER 2022 and 2021 - (UNAUDITED)
- 29 -
WORLDWIDE UNITED STATES
TOTAL INTERNATIONAL
(a)
2022 2021
% Change
2022 2021
% Change
2022 2021
% Change
(MILLIONS)
Total Oper. Total Total Oper.
TOTAL REVENUES
$ 100,330
$ 81,288 23% 30% $ 42,473
$ 29,746
43% $ 57,857 $ 51,542 12% 23%
GLOBAL BIOPHARMACEUTICALS BUSINESS (BIOPHARMA)
(b)
$ 98,988 $ 79,557 24% 31% $ 42,083
$ 29,221
44% $ 56,905 $ 50,336 13% 24%
Primary Care $ 73,023 $ 52,029 40% 49% $ 28,503
$ 15,329
86% $ 44,521 $ 36,700 21% 33%
Comirnaty direct sales and alliance revenues 37,806 36,781 3% 10% 8,775 7,809 12% 29,032 28,972 — 9%
Paxlovid 18,933 76 * * 10,514 76 * 8,419 — * *
Eliquis alliance revenues and direct sales 6,480 5,970 9% 14% 3,822 3,160 21% 2,658 2,810 (5%) 5%
Prevnar family
(c)
6,337 5,272 20% 23% 4,032 2,701 49% 2,305 2,571 (10%) (4%)
Premarin family 455 563 (19%) (19%) 420 525 (20%) 36 38 (6%) 1%
BMP2 277 266 4% 4% 277 266 4% — — — —
Nimenrix 268 193 39% 52% — — — 268 193 39% 52%
Nurtec ODT/Vydura 213 — * * 211 — * 2 — * *
FSME-IMMUN/TicoVac 200 185 8% 20% 2 — * 198 185 7% 19%
Toviaz 146 238 (39%) (32%) 27 68 (61%) 119 170 (30%) (20%)
Trumenba 123 118 4% 5% 111 106 5% 12 13 (7%) 4%
Chantix/Champix 8 398 (98%) (98%) 8 310 (97%) — 88 * *
All other Primary Care 1,778 1,967 (10%) (2%) 305 308 (1%) 1,473 1,659 (11%) (2%)
Specialty Care $ 13,833 $ 15,194 (9%) (4%) $ 5,659 $ 6,156 (8%) $ 8,174 $ 9,038 (10%) (1%)
Vyndaqel family
(d)
2,447 2,015 21% 29% 1,245 909 37% 1,202 1,106 9% 22%
Xeljanz 1,796 2,455 (27%) (24%) 1,129 1,647 (31%) 668 808 (17%) (8%)
Enbrel (Outside the U.S. and Canada) 1,003 1,185 (15%) (6%) — — — 1,003 1,185 (15%) (6%)
Sulperazon 786 683 15% 19% — — — 786 683 15% 19%
Inflectra 532 657 (19%) (16%) 289 385 (25%) 243 272 (11%) (4%)
Ig Portfolio
(e)
491 430 14% 14% 491 430 14% — — — —
BeneFIX 425 438 (3%) 3% 235 230 2% 191 207 (8%) 5%
Zavicefta 412 413 — 8% — — — 412 413 — 8%
Genotropin 360 389 (8%) 4% 68 79 (14%) 292 310 (6%) 8%
Zithromax 331 278 19% 26% 2 1 * 328 278 18% 25%
Medrol 328 432 (24%) (20%) 132 181 (27%) 196 251 (22%) (15%)
Fragmin 269 305 (12%) (4%) 4 5 (22%) 264 300 (12%) (4%)
Somavert 268 277 (4%) 3% 111 98 13% 157 179 (12%) (2%)
Refacto AF/Xyntha 239 304 (21%) (14%) 59 64 (8%) 180 240 (25%) (15%)
Vfend 225 267 (16%) (10%) 5 8 (37%) 221 260 (15%) (9%)
Oxbryta 73 — * * 72 — * — — — —
All other Anti-infectives 1,471 1,835 (20%) (15%) 454 511 (11%) 1,017 1,324 (23%) (16%)
All other Specialty Care 2,377 2,830 (16%) (13%) 1,362 1,608 (15%) 1,015 1,223 (17%) (11%)
Oncology $ 12,132 $ 12,333 (2%) 2% $ 7,921 $ 7,736 2% $ 4,210 $ 4,597 (8%) 1%
Ibrance 5,120 5,437 (6%) (2%) 3,370 3,418 (1%) 1,751 2,019 (13%) (4%)
Xtandi alliance revenues 1,198 1,185 1% 1% 1,198 1,185 1% — — — —
Inlyta 1,003 1,002 — 4% 618 599 3% 385 403 (5%) 5%
Bosulif 575 540 6% 11% 375 354 6% 200 186 7% 21%
Zirabev 562 444 27% 31% 413 246 68% 149 197 (25%) (15%)
Xalkori 465 493 (6%) (1%) 105 102 3% 361 391 (8%) (2%)
Ruxience 458 491 (7%) (6%) 403 450 (10%) 55 41 34% 47%
Retacrit 394 444 (11%) (9%) 312 344 (9%) 82 100 (18%) (8%)
Sutent 347 673 (48%) (45%) 33 127 (74%) 314 546 (42%) (38%)
Lorbrena 343 266 29% 37% 177 141 26% 166 125 33% 50%
Bavencio alliance revenues 271 178 52% 65% 101 83 22% 169 95 79% *
Aromasin 248 211 17% 23% 2 3 (26%) 246 208 18% 24%
Besponsa 219 192 14% 20% 126 115 10% 93 77 21% 36%
Trazimera 203 197 3% 7% 106 98 8% 97 99 (2%) 5%
Braftovi 194 187 4% 4% 191 187 2% 3 — * *
Mektovi 176 155 14% 14% 175 155 13% 1 — * *
All other Oncology 357 238 50% 54% 216 129 68% 140 109 28% 38%
PFIZER CENTREONE
(b)
$ 1,342 $ 1,731 (22%) (19%) $ 390 $ 524 (26%) $ 952 $ 1,206 (21%) (16%)
Total Alliance revenues included above $ 8,537 $ 7,652 12% 15% $ 5,203 $ 4,456 17% $ 3,335 $ 3,195 4% 12%
PFIZER INC. - REVENUES
TWELVE MONTHS 2022 and 2021 - (UNAUDITED)
- 30 -

DEVELOPED EUROPE
(f)
DEVELOPED REST OF WORLD
(g)
EMERGING MARKETS
(h)
2022 2021
% Change
2022 2021
% Change
2022 2021
% Change
(MILLIONS)
Total Oper. Total Oper. Total Oper.
TOTAL INTERNATIONAL REVENUES
$ 21,982
$ 18,336
20% 33% $ 15,778 $ 12,506 26% 43%
$ 20,097
$ 20,701
(3%) 2%
GLOBAL BIOPHARMACEUTICALS BUSINESS (BIOPHARMA)
(b)
$ 21,260
$ 17,662
20% 34% $ 15,692 $ 12,399 27% 44%
$ 19,954
$ 20,275
(2%) 3%
Primary Care
$ 16,882
$ 12,629
34% 48% $ 13,137 $ 9,413 40% 59%
$ 14,502
$ 14,658
(1%) 3%
Comirnaty direct sales and alliance revenues 10,827 9,410 15% 27% 8,330 8,143 2% 18% 9,875 11,419 (14%) (11%)
Paxlovid 3,237 — * * 3,732 — * * 1,450 — * *
Eliquis alliance revenues and direct sales 1,459 1,520 (4%) 8% 407 439 (7%) 5% 793 852 (7%) 1%
Prevnar family
(c)
512 655 (22%) (12%) 337 388 (13%) (2%) 1,456 1,528 (5%) —
Premarin family 1 1 (13%) (4%) 19 20 (3%) 5% 15 17 (9%) (4%)
BMP2 — — — — — — — — — — — —
Nimenrix 96 132 (28%) (19%) 19 16 16% 24% 154 45 * *
Nurtec ODT/Vydura — — — — — — — — 2 — * *
FSME-IMMUN/TicoVac 160 154 4% 17% — — — — 37 31 21% 33%
Toviaz 42 72 (42%) (36%) 70 89 (21%) (10%) 7 10 (24%) 5%
Trumenba 9 11 (21%) (11%) — — — — 3 1 * *
Chantix/Champix — 46 (99%) (99%) (1) 30 * * — 12 (99%) (99%)
All other Primary Care 539 627 (14%) (5%) 223 289 (23%) (13%) 710 744 (5%) 4%
Specialty Care $ 2,643 $ 2,886 (8%) 2% $ 1,683 $ 2,060 (18%) (9%) $ 3,849 $ 4,092 (6%) —
Vyndaqel family
(d)
821 572 43% 61% 308 495 (38%) (29%) 73 39 90% 100%
Xeljanz 235 308 (24%) (15%) 232 284 (18%) (8%) 200 216 (7%) 1%
Enbrel (Outside the U.S. and Canada) 394 533 (26%) (17%) 195 251 (22%) (11%) 414 401 3% 13%
Sulperazon — — — — 4 7 (41%) (31%) 782 677 16% 19%
Inflectra 120 186 (35%) (28%) 112 75 49% 55% 11 11 (3%) 10%
Ig Portfolio
(e)
— — — — — — — — — — — —
BeneFIX 54 70 (23%) (14%) 52 58 (10%) 1% 84 79 7% 25%
Zavicefta 103 126 (18%) (8%) 1 1 (1%) 8% 307 286 7% 15%
Genotropin 103 119 (13%) (3%) 89 105 (15%) (2%) 100 86 15% 35%
Zithromax 49 44 10% 24% 19 21 (10%) 4% 261 212 23% 27%
Medrol 56 61 (7%) 4% 36 43 (17%) (8%) 103 147 (30%) (25%)
Fragmin 144 155 (7%) 3% 53 55 (5%) (2%) 68 89 (24%) (19%)
Somavert 120 136 (12%) (1%) 19 24 (20%) (13%) 18 20 (8%) 4%
Refacto AF/Xyntha 77 119 (35%) (28%) 16 23 (31%) (25%) 87 98 (11%) 2%
Vfend 13 21 (37%) (29%) 39 44 (11%) 4% 168 195 (14%) (10%)
Oxbryta — — — — — — — — — — — —
All other Anti-infectives 261 292 (11%) — 99 115 (14%) (3%) 657 916 (28%) (23%)
All other Specialty Care 92 145 (36%) (30%) 409 459 (11%) (4%) 514 620 (17%) (12%)
Oncology $ 1,735 $ 2,148 (19%) (10%) $ 872 $ 925 (6%) 7% $ 1,603 $ 1,525 5% 12%
Ibrance 845 1,044 (19%) (10%) 408 453 (10%) 2% 498 522 (5%) 4%
Xtandi alliance revenues — — — — — — — — — — — —
Inlyta 153 181 (16%) (5%) 72 91 (22%) (10%) 160 131 23% 29%
Bosulif 97 92 6% 18% 66 65 2% 19% 36 29 24% 34%
Zirabev 100 149 (33%) (25%) 38 38 1% 14% 11 11 3% 29%
Xalkori 82 94 (13%) (3%) 38 46 (18%) (7%) 241 251 (4%) —
Ruxience 21 18 16% 31% 25 19 29% 39% 9 4 * *
Retacrit 79 98 (19%) (8%) — — — — 3 2 11% 17%
Sutent 68 200 (66%) (63%) 53 75 (29%) (20%) 193 271 (29%) (24%)
Lorbrena 66 55 19% 34% 39 40 (4%) 11% 61 29 * *
Bavencio alliance revenues 79 53 50% 69% 64 34 87% * 26 8 * *
Aromasin 24 28 (15%) (5%) 6 8 (33%) (22%) 216 172 26% 31%
Besponsa 35 30 18% 32% 28 32 (11%) 2% 30 16 89% *
Trazimera 37 46 (18%) (9%) 8 8 (1%) 9% 51 45 13% 18%
Braftovi — — — — 2 — * * — — — —
Mektovi — — — — 1 — * * — — — —
All other Oncology 49 60 (19%) (10%) 24 15 66% 77% 67 35 94% *
PFIZER CENTREONE
(b)
$ 722 $ 673 7% 14% $ 86 $ 107 (19%) (7%) $ 143 $ 426 (66%) (66%)
Total Alliance revenues included above $ 2,749 $ 2,248 22% 30% $ 489 $ 496 (1%) 12% $ 97 $ 451 (78%) (78%)
PFIZER INC.
INTERNATIONAL REVENUES BY GEOGRAPHIC REGION
TWELVE MONTHS 2022 and 2021 - (UNAUDITED)
- 31 -
PFIZER INC.
NOTES TO REVENUES TABLE INFORMATION
(UNAUDITED)
(a)
Total International represents Developed Europe region + Developed Rest of World region + Emerging Markets region. Details for these regions are described in footnotes
(f) to (h) below, respectively.
(b)
We manage our commercial operations through two operating segments, each led by a single manager: Biopharma, our innovative science-based biopharmaceutical
business, and Pfizer CentreOne (PC1), our global contract development and manufacturing organization and a leading supplier of specialty active pharmaceutical
ingredients. Beginning in the third quarter of 2022, we made several organizational changes to further transform our operations to better leverage our expertise in certain
areas and in anticipation of potential future new product or indication launches. The changes included establishing a new commercial structure within Biopharma to better
support and optimize performance across three broad customer groups:
• Primary Care consists of the former Internal Medicine and Vaccines product portfolios, products for COVID-19 prevention and treatment, and potential future mRNA
and antiviral products.
• Specialty Care consists of the former Inflammation & Immunology, Rare Disease and Hospital (excluding Paxlovid) product portfolios.
• Oncology consists of the former Oncology product portfolio.
Prior-period financial information has been revised to reflect the current period presentation.
PC1 includes revenues from our contract manufacturing, including certain Comirnaty-related manufacturing activities performed on behalf of BioNTech ($80 million and
$188 million for the fourth quarter and the twelve months of 2022, respectively, and $46 million and $320 million for the fourth quarter and the twelve months of 2021,
respectively), and revenues from our active pharmaceutical ingredient sales operation, as well as revenues related to our manufacturing and supply agreements with former
legacy Pfizer businesses/partnerships, including but not limited to, transitional manufacturing and supply agreements with Viatris following the spin-off of the Upjohn
Business.
(c)
Prevnar family includes revenues from Prevnar 13/Prevenar 13 (pediatric and adult) and Prevnar 20/Apexxnar (adult).
(d)
Vyndaqel family includes global revenues from Vyndaqel, as well as revenues for Vyndamax in the U.S. and Vynmac in Japan.
(e)
Immunoglobulin (Ig) portfolio includes the revenues from Panzyga, Octagam and Cutaquig.
(f)
Developed Europe region includes the following markets: Western Europe, Scandinavian countries and Finland.
(g)
Developed Rest of World region includes the following markets: Japan, Canada, South Korea, Australia and New Zealand.
(h)
Emerging Markets region includes, but is not limited to, the following markets: Asia (excluding Japan and South Korea), Latin America, Central Europe, Eastern Europe,
the Middle East, Africa and Turkey.
* Indicates calculation not meaningful.
Amounts may not add due to rounding. All percentages have been calculated using unrounded amounts.
- 32 -

DISCLOSURE NOTICE: Except where otherwise noted, the information contained in this earnings release and the related
attachments is as of January 31, 2023. We assume no obligation to update any forward-looking statements contained in this
earnings release and the related attachments as a result of new information or future events or developments.
This earnings release and the related attachments contain forward-looking statements about, among other topics, our
anticipated operating and financial performance; reorganizations; business plans, strategy and prospects; our
Environmental, Social and Governance (ESG) priorities, strategy and goals; expectations for our product pipeline, in-line
products and product candidates, including anticipated regulatory submissions, data read-outs, study starts, approvals,
launches, clinical trial results and other developing data, revenue contribution and projections, pricing and reimbursement,
potential market dynamics and size, growth, performance, timing of exclusivity and potential benefits; strategic reviews;
capital allocation objectives; dividends and share repurchases; plans for and prospects of our acquisitions, dispositions and
other business development activities, and our ability to successfully capitalize on these opportunities; manufacturing and
product supply; our ongoing efforts to respond to COVID-19, including the Pfizer-BioNTech COVID-19 Vaccine
(Comirnaty), the Pfizer-BioNTech COVID-19 Omicron BA.4/BA.5 Vaccine, Bivalent (the Pfizer-BioNTech COVID-19
bivalent vaccine), other vaccines that may result from the BNT162 program, including new variant-based or next-
generation vaccines, and our oral COVID-19 treatment (Paxlovid); and our expectations regarding the impact of
COVID-19 on our business, operations and financial results that involve substantial risks and uncertainties. You can
identify these statements by the fact that they use future dates or use words such as “will,” “may,” “could,” “likely,”
“ongoing,” “anticipate,” “estimate,” “expect,” “project,” “intend,” “plan,” “believe,” “assume,” “target,” “forecast,”
“guidance,” “goal,” “objective,” “aim,” “seek,” “potential,” “hope” and other words and terms of similar meaning.
Among the factors that could cause actual results to differ materially from past results and future plans and projected future
results are the following:
Risks Related to Our Business, Industry and Operations, and Business Development:
• the outcome of research and development (R&D) activities, including, the ability to meet anticipated pre-clinical or
clinical endpoints, commencement and/or completion dates for our pre-clinical or clinical trials, regulatory submission
dates, and/or regulatory approval and/or launch dates; the possibility of unfavorable pre-clinical and clinical trial results,
including the possibility of unfavorable new pre-clinical or clinical data and further analyses of existing pre-clinical or
clinical data; risks associated with preliminary, early stage or interim data; the risk that pre-clinical and clinical trial data
are subject to differing interpretations and assessments, including during the peer review/publication process, in the
scientific community generally, and by regulatory authorities; and whether and when additional data from our pipeline
programs will be published in scientific journal publications and, if so, when and with what modifications and
interpretations;
• our ability to successfully address comments received from regulatory authorities such as the FDA or the EMA, or obtain
approval for new products and indications from regulators on a timely basis or at all; regulatory decisions impacting
labeling, including the scope of indicated patient populations, product dosage, manufacturing processes, safety and/or
other matters, including decisions relating to emerging developments regarding potential product impurities; the impact
of, or uncertainties regarding the ability to obtain, recommendations by technical or advisory committees; and the timing
of pricing approvals and product launches;
• claims and concerns that may arise regarding the safety or efficacy of in-line products and product candidates, including
claims and concerns that may arise from the outcome of post-approval clinical trials, which could impact marketing
approval, product labeling, and/or availability or commercial potential, including uncertainties regarding the commercial
or other impact of the results of the Xeljanz ORAL Surveillance (A3921133) study or actions by regulatory authorities
based on analysis of ORAL Surveillance or other data, including on other Janus kinase (JAK) inhibitors in our portfolio;
• the success and impact of external business development activities, including the ability to identify and execute on
potential business development opportunities; the ability to satisfy the conditions to closing of announced transactions in
the anticipated time frame or at all; the ability to realize the anticipated benefits of any such transactions in the
anticipated time frame or at all; the potential need for and impact of additional equity or debt financing to pursue these
opportunities, which could result in increased leverage and/or a downgrade of our credit ratings; challenges integrating
the businesses and operations; disruption to business and operations relationships; risks related to growing revenues for
certain acquired products; significant transaction costs; and unknown liabilities;
• competition, including from new product entrants, in-line branded products, generic products, private label products,
biosimilars and product candidates that treat or prevent diseases and conditions similar to those treated or intended to be
prevented by our in-line products and product candidates;
• the ability to successfully market both new and existing products, including biosimilars;
• difficulties or delays in manufacturing, sales or marketing; supply disruptions, shortages or stock-outs at our facilities or
third-party facilities that we rely on; and legal or regulatory actions;
- 33 -
• the impact of public health outbreaks, epidemics or pandemics (such as the COVID-19 pandemic) on our business,
operations and financial condition and results, including impacts on our employees, manufacturing, supply chain, sales
and marketing, research and development and clinical trials;
• risks and uncertainties related to our efforts to develop and commercialize our COVID-19 products, as well as challenges
related to their manufacturing, supply and distribution, including, among others, uncertainties inherent in research and
development, including the ability to meet anticipated clinical endpoints, commencement and/or completion dates for
clinical trials, regulatory submission dates, regulatory approval dates and/or launch dates, as well as risks associated with
pre-clinical and clinical data (including Phase 1/2/3 or Phase 4 data for Comirnaty, the Pfizer-BioNTech COVID-19
bivalent vaccine, any monovalent, bivalent or variant-adapted vaccine candidates or any other vaccine candidate in the
BNT162 program or Paxlovid or any future COVID-19 treatment) in any of our studies in pediatrics, adolescents or
adults or real world evidence, including the possibility of unfavorable new pre-clinical, clinical or safety data and further
analyses of existing pre-clinical, clinical or safety data or further information regarding the quality of pre-clinical,
clinical or safety data, including by audit or inspection; the ability to produce comparable clinical or other results for
Comirnaty, the Pfizer-BioNTech COVID-19 bivalent vaccine, any monovalent, bivalent or variant-adapted vaccine
candidates or other vaccines that may result from the BNT162 program, Paxlovid or any future COVID-19 treatment or
any other COVID-19 program, including the rate of effectiveness and/or efficacy, safety and tolerability profile observed
to date, in additional analyses of the Phase 3 trial for any such products and additional studies, in real-world data studies
or in larger, more diverse populations following commercialization; the ability of Comirnaty, the Pfizer-BioNTech
COVID-19 bivalent vaccine, any monovalent, bivalent or variant-adapted vaccine candidates or any future vaccine to
prevent, or Paxlovid or any future COVID-19 treatment to be effective against, COVID-19 caused by emerging virus
variants; the risk that more widespread use of Comirnaty, the Pfizer-BioNTech COVID-19 bivalent vaccine or Paxlovid
will lead to new information about efficacy, safety or other developments, including the risk of additional adverse
reactions, some of which may be serious; the risk that pre-clinical and clinical trial data are subject to differing
interpretations and assessments, including during the peer review/publication process, in the scientific community
generally, and by regulatory authorities; whether and when additional data from the BNT162 mRNA vaccine program,
Paxlovid or other COVID-19 programs will be published in scientific journal publications and, if so, when and with what
modifications and interpretations; whether regulatory authorities will be satisfied with the design of and results from
existing or future pre-clinical and clinical studies; whether and when submissions to request emergency use or
conditional marketing authorizations for Comirnaty, the Pfizer-BioNTech COVID-19 bivalent vaccine, or any future
vaccines in additional populations, for a potential booster dose for Comirnaty, the Pfizer-BioNTech COVID-19 bivalent
vaccine, any monovalent or bivalent vaccine candidates or any potential future vaccines (including potential future
annual boosters or re-vaccinations), and/or biologics license and/or EUA applications or amendments to any such
applications may be filed in particular jurisdictions for Comirnaty, the Pfizer-BioNTech COVID-19 bivalent vaccine,
any monovalent or bivalent vaccine candidates or any other potential vaccines that may arise from the BNT162 program,
including a potential variant-based, higher dose, or bivalent vaccine or any other potential vaccines, and if obtained,
whether or when such EUA or licenses will expire or terminate; whether and when submissions to request emergency
use or conditional marketing authorizations for Paxlovid or any future COVID-19 treatment and/or any drug applications
and/or EUA applications or amendments to any such applications for any indication for Paxlovid or any future
COVID-19 treatment may be filed in particular jurisdictions, and if obtained, whether or when such EUA or licenses will
expire or terminate; whether and when any application that may be pending or filed for Comirnaty, the Pfizer-BioNTech
COVID-19 bivalent vaccine, any monovalent, bivalent or variant-adapted vaccine candidates or other vaccines that may
result from the BNT162 program, Paxlovid or any future COVID-19 treatment or any other COVID-19 program may be
approved by particular regulatory authorities, which will depend on myriad factors, including making a determination as
to whether the vaccine’s or drug’s benefits outweigh its known risks and determination of the vaccine’s or drug’s
efficacy and, if approved, whether it will be commercially successful; decisions by regulatory authorities impacting
labeling or marketing, manufacturing processes, safety and/or other matters that could affect the availability or
commercial potential of a vaccine or drug, including the authorization or approval of products or therapies developed by
other companies; disruptions in the relationships between us and our collaboration partners, clinical trial sites or third-
party suppliers, including our relationship with BioNTech; the risk that other companies may produce superior or
competitive products; the risk that demand for any products may be reduced, no longer exist or not meet expectations
which may lead to excess inventory on-hand and/or in the channel or reduced revenues; challenges related to a transition
to the commercial market for any of the products; risks related to the availability of raw materials to manufacture or test
any such products; challenges related to our vaccine’s formulation, dosing schedule and attendant storage, distribution
and administration requirements, including risks related to storage and handling after delivery by Pfizer; challenges and
risks related to medication errors such as prescribing or dispensing the wrong strength, improper dosing and self-
administration errors; the risk that we may not be able to successfully develop other vaccine formulations, booster doses
or potential future annual boosters or re-vaccinations or new variant-based or next generation vaccines or next generation
COVID-19 treatments; uncertainties related to vaccine adherence; the risk that we may not be able to recoup costs
- 34 -

associated with our R&D and manufacturing efforts; risks associated with any changes in the way we approach or
provide research funding for the BNT162 program, Paxlovid or any other COVID-19 program; challenges and risks
associated with the pace of our development programs; the risk that we may not be able to maintain manufacturing
capacity or access to logistics or supply channels commensurate with global demand for our COVID-19 products, which
would negatively impact our ability to supply our COVID-19 products within the projected time periods; risks related to
our ability to achieve our revenue forecasts for Comirnaty, the Pfizer-BioNTech COVID-19 bivalent vaccine and
Paxlovid or any potential future COVID-19 vaccines or treatments; whether and when additional supply or purchase
agreements will be reached or existing agreements will be completed; uncertainties regarding the ability to obtain
recommendations from vaccine or treatment advisory or technical committees and other public health authorities and
uncertainties regarding the commercial impact of any such recommendations; pricing and access challenges for such
products; challenges related to public confidence in, or awareness of Comirnaty, the Pfizer-BioNTech COVID-19
bivalent vaccine or Paxlovid, including challenges driven by misinformation or disinformation, access, concerns about
clinical data integrity, or prescriber and pharmacy education; trade restrictions; potential third-party royalties or other
claims related to Comirnaty or Paxlovid; and competitive developments;
• trends toward managed care and healthcare cost containment, and our ability to obtain or maintain timely or adequate
pricing or favorable formulary placement for our products;
• interest rate and foreign currency exchange rate fluctuations, including the impact of possible currency devaluations in
countries experiencing high inflation rates;
• any significant issues involving our largest wholesale distributors or government customers, which account for a
substantial portion of our revenues;
• the impact of the increased presence of counterfeit medicines or vaccines in the pharmaceutical supply chain;
• any significant issues related to the outsourcing of certain operational and staff functions to third parties; and any
significant issues related to our JVs and other third-party business arrangements;
• uncertainties related to general economic, political, business, industry, regulatory and market conditions including,
without limitation, uncertainties related to the impact on us, our customers, suppliers and lenders and counterparties to
our foreign-exchange and interest-rate agreements of challenging global economic conditions, such as inflation, and
recent and possible future changes in global financial markets;
• any changes in business, political and economic conditions due to actual or threatened terrorist activity, geopolitical
instability, civil unrest or military action;
• the impact of product recalls, withdrawals and other unusual items, including uncertainties related to regulator-directed
risk evaluations and assessments, including our ongoing evaluation of our product portfolio for the potential presence or
formation of nitrosamines;
• trade buying patterns;
• the risk of an impairment charge related to our intangible assets, goodwill or equity-method investments;
• the impact of, and risks and uncertainties related to, restructurings and internal reorganizations, as well as any other
corporate strategic initiatives and growth strategies, and cost-reduction and productivity initiatives, each of which
requires upfront costs but may fail to yield anticipated benefits and may result in unexpected costs or organizational
disruption;
• the ability to successfully achieve our climate goals and progress our environmental sustainability priorities;
Risks Related to Government Regulation and Legal Proceedings:
• the impact of any U.S. healthcare reform or legislation or any significant spending reductions or cost controls affecting
Medicare, Medicaid or other publicly funded or subsidized health programs, including the Inflation Reduction Act of
2022, or changes in the tax treatment of employer-sponsored health insurance that may be implemented;
• U.S. federal or state legislation or regulatory action and/or policy efforts affecting, among other things, pharmaceutical
product pricing, intellectual property, reimbursement or access or restrictions on U.S. direct-to-consumer advertising;
limitations on interactions with healthcare professionals and other industry stakeholders; as well as pricing pressures for
our products as a result of highly competitive insurance markets;
• legislation or regulatory action in markets outside of the U.S., including China, affecting pharmaceutical product pricing,
intellectual property, reimbursement or access, including, in particular, continued government-mandated reductions in
prices and access restrictions for certain biopharmaceutical products to control costs in those markets;
• the exposure of our operations globally to possible capital and exchange controls, economic conditions, expropriation
and other restrictive government actions, changes in intellectual property legal protections and remedies, the impact of
political or civil unrest or military action, including the ongoing conflict between Russia and Ukraine and its economic
- 35 -

consequences, unstable governments and legal systems, inter-governmental disputes and natural disasters or disruptions
related to climate change;
• legal defense costs, insurance expenses, settlement costs and contingencies, including those related to actual or alleged
environmental contamination;
• the risk and impact of an adverse decision or settlement and the risk related to adequacy of reserves related to legal
proceedings;
• the risk and impact of tax related litigation and investigations;
• governmental laws and regulations affecting our operations, including, without limitation, the recently enacted Inflation
Reduction Act of 2022, changes in laws and regulations or their interpretation, including, among others, changes in tax
laws and regulations internationally and in the U.S., the adoption of global minimum taxation requirements outside the
U.S. and potential changes to existing tax law by the current U.S. Presidential administration and Congress;
Risks Related to Intellectual Property, Technology and Security:
• any significant breakdown or interruption of our information technology systems and infrastructure (including cloud
services);
• any business disruption, theft of confidential or proprietary information, security threats on facilities or infrastructure,
extortion or integrity compromise resulting from a cyber-attack or other malfeasance by, but not limited to, nation states,
employees, business partners or others;
• the risk that our currently pending or future patent applications may not be granted on a timely basis or at all, or any
patent-term extensions that we seek may not be granted on a timely basis, if at all; and
• our ability to protect our products, patents and other intellectual property, such as: (i) against claims of invalidity that
could result in loss of exclusivity; (ii) claims of patent infringement; (iii) challenges faced by our collaboration or
licensing partners to the validity of their patent rights; and (iv) in response to any pressure, or legal or regulatory action
by, various stakeholders or governments that could potentially result in us not seeking intellectual property protection or
agreeing not to enforce or being restricted from enforcing intellectual property rights related to our products, including
Comirnaty, the Pfizer-BioNTech COVID-19 bivalent vaccine and Paxlovid.
We cannot guarantee that any forward-looking statement will be realized. Should known or unknown risks or uncertainties
materialize or should underlying assumptions prove inaccurate, actual results could vary materially from past results and
those anticipated, estimated or projected. Investors are cautioned not to put undue reliance on forward-looking statements.
A further list and description of risks, uncertainties and other matters can be found in our Annual Report on Form 10-K for
the fiscal year ended December 31, 2021 and in our subsequent report on Form 10-Q, in each case including in the sections
thereof captioned “Forward-Looking Information and Factors That May Affect Future Results” and “Item 1A. Risk
Factors,” and in our subsequent reports on Form 8-K.
This earnings release may include discussion of certain clinical studies relating to various in-line products and/or product
candidates. These studies typically are part of a larger body of clinical data relating to such products or product candidates,
and the discussion herein should be considered in the context of the larger body of data. In addition, clinical trial data are
subject to differing interpretations, and, even when we view data as sufficient to support the safety and/or effectiveness of a
product candidate or a new indication for an in-line product, regulatory authorities may not share our views and may
require additional data or may deny approval altogether.
The information contained on our website or any third-party website is not incorporated by reference into this earnings
release. All trademarks mentioned are the property of their owners.
- 36 -
